Biological therapies (immunotherapies)
Biological therapy is a cancer treatment that uses the body’s immune system to destroy cancer cells.
The information provided on www.fcarreras.org is intended to support, not replace, the relationship that exists between patients/visitors to this website and their physician.
What is a biological therapy or immunotherapy and what is its purpose?
 Biological therapies or immunotherapies use elements of the body’s own immune system to attack cancer. Some of these therapies are very new and there is currently a lot of research ongoing in this area. In some cases these are treatments still being researched, therefore some of the biologic therapy options may only be accessible through clinical trials.
Biological therapies or immunotherapies use elements of the body’s own immune system to attack cancer. Some of these therapies are very new and there is currently a lot of research ongoing in this area. In some cases these are treatments still being researched, therefore some of the biologic therapy options may only be accessible through clinical trials.
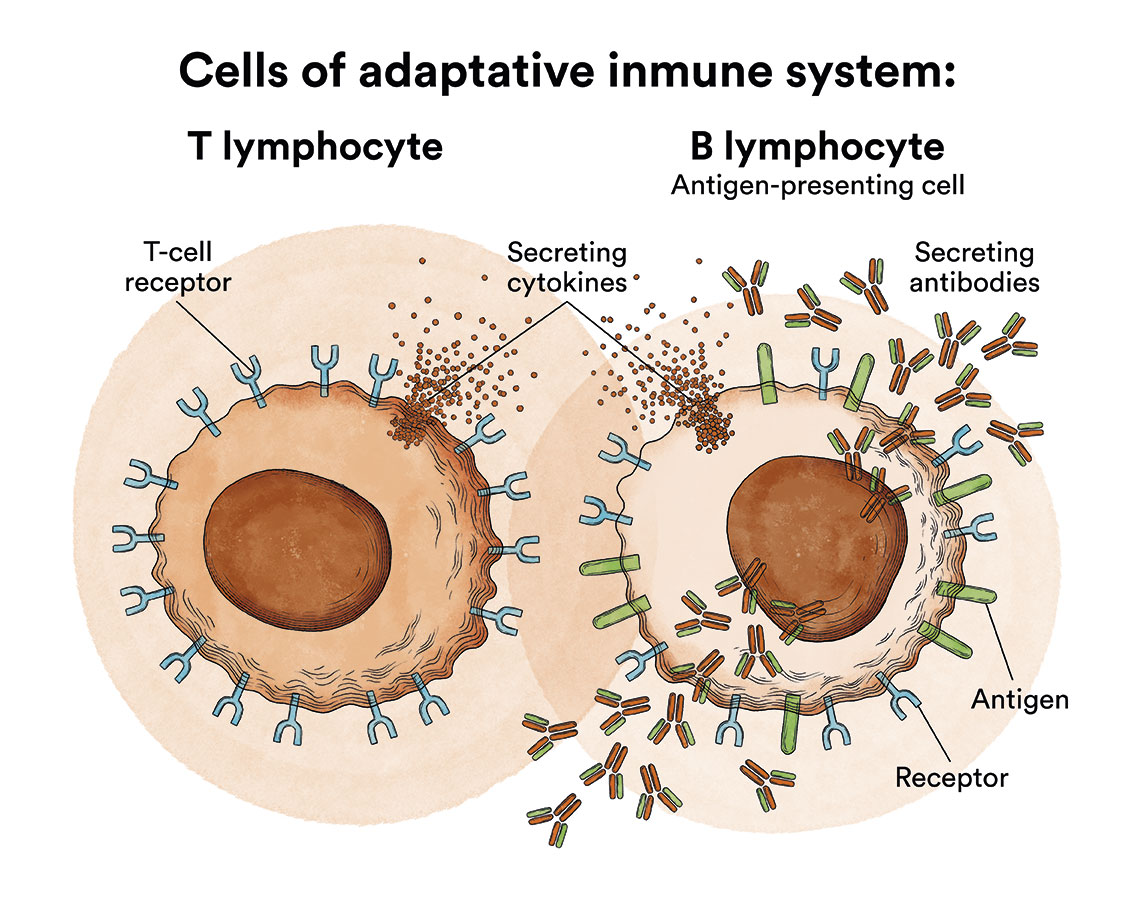
Our body’s immune system has the ability to recognise and attack foreign elements. It is a complex network of cells, tissues and organs that together help the body fight infection and other diseases. When germs such as bacteria or viruses invade the body, they attack and multiply. The immune system defends our body against substances it considers harmful or foreign. These substances are called antigens. When the immune system recognises an antigen, it attacks it. This is called an immune response. Part of this response is to produce antibodies. Antibodies are proteins that act to attack, weaken and destroy antigens.
One of the most important elements of this immune system are the lymphocytes. These are a type of white blood cell made in the bone marrow; these are located in the blood and lymphoid tissue. The two main types of lymphocytes are B-lymphocytes and T-lymphocytes. B-lymphocytes make antibodies and T-lymphocytes help destroy tumour cells and control immune responses.
These lymphocytes are created in the bone marrow (See Leukaemia, Bone Marrow and Blood Cells), the body’s ‘blood factory’. Bone marrow is a spongy tissue found inside some of the bones of the body such as the iliac crests (hip bone), the sternum or the bones forming the skull. In colloquial language it is called marrow. It should not be mistaken for the spinal cord as they have nothing to do with each other. Their functions are totally different. The spinal cord is located in the spinal column and transmits nerve impulses from the brain to the entire body and vice versa.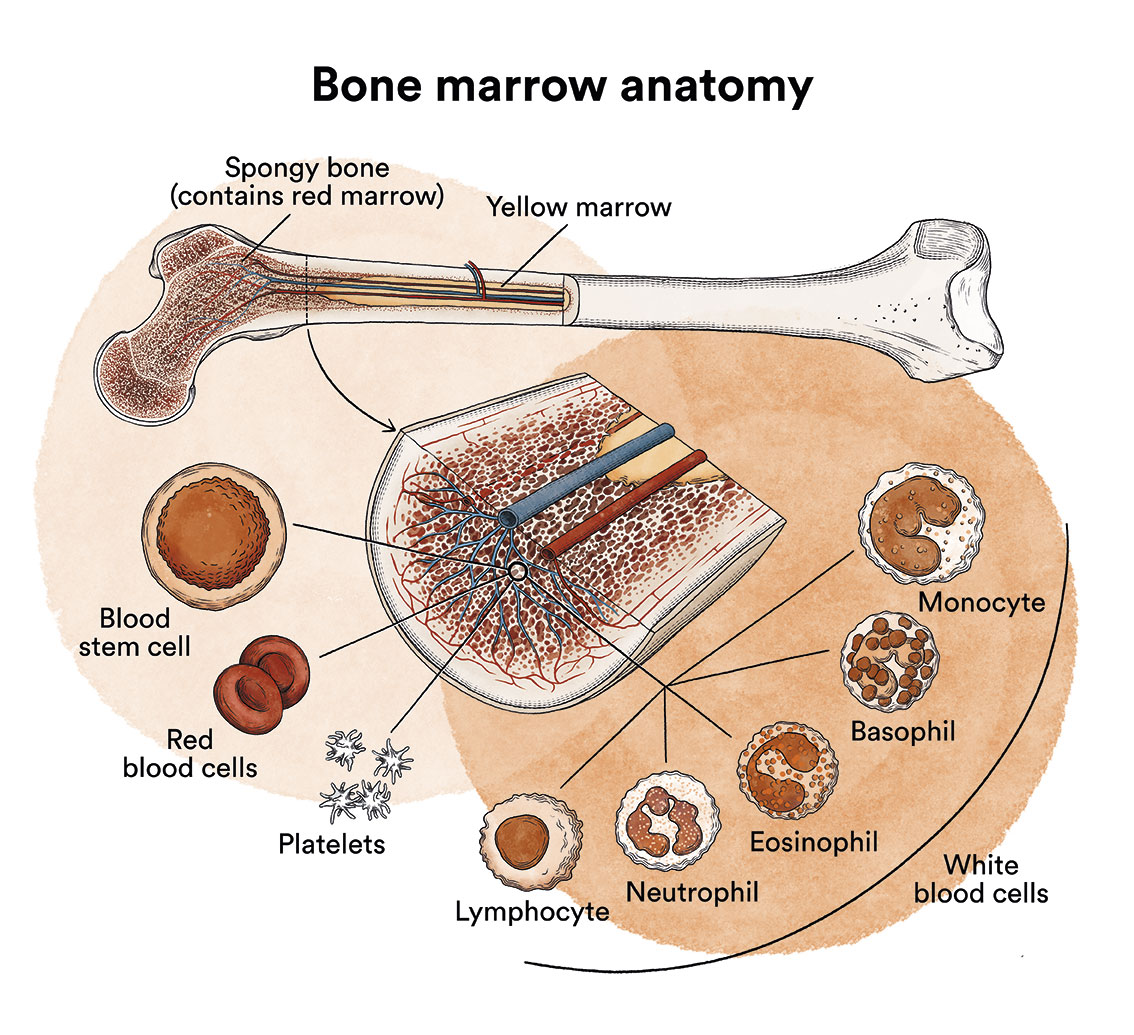
Biological therapies aim to get the immune system to detect and try to eliminate cancer cells. However, cancer cells can sometimes camouflage themselves or even disable or inhibit the immune system so that it does not act. For example, some cancer cells have genetic mutations that make them less visible to the immune system, some have proteins on their surface that cause immune system cells to ‘switch off’, or others create a microclimate around the tumour that interferes with the effectiveness of the immune system. So there are biological therapies that trigger the immune system to act and others that also make these cancer cells “visible”.
What types of biological therapies or immunotherapies are there and how do they work?
Let’s look at some of the main types of biological therapies that can be used in the treatment of leukaemia, lymphomas or other blood cancers:
Monoclonal antibodies
As mentioned above, the B-lymphocytes in our body make antibodies for the immune system to fight against foreign substances, the antigens. An antibody is a protein that circulates in the body and, when it encounters an antigen, the former binds to it. These and other immune system cells present these antigens and activate other immune system cells to attack them.
A monoclonal antibody is a man-made protein that acts as an antibody created and designed in the laboratory to attack a particular antigen expressed by the cancer cell.
At a research level, antigens specific to some cancer cells have been detected. Therefore, different types of monoclonal antibodies have been and are being developed. When we talk about monoclonal antibodies, we talk about targeted therapies because they have a specific target on the cancer cell they find; they bind to the specific antigen on the cancer cell so that the cancer cell can eventually be eliminated.
Monoclonal antibodies are known as mAbs, which is why their drugs have names ending in -mab. Some are called “bispecific”, as they are composed of parts of two different mAbs, meaning that they can bind to two different proteins at the same time.
Examples include brentuximab, an antibody directed against the CD30 molecule and approved for some types of lymphomas such as Hodgkin’s; or blinatumomab, directed against CD19 and CD3, and used in some types of acute leukaemia.
More info:
– Monoclonal antibodies and their side effects. National Cancer Institute
CAR-T immunotherapy (or CAR-T cells)
A T-lymphocyte is a cell in our own body, a type of white blood cell created by our bone marrow. T-lymphocytes, which develop in the thymus, are actively involved in the body’s cell-mediated immune response. T-lymphocytes have a special receptor on the cell membrane that is specific to them, called the T-cell receptor.

Tumour cells express receptors on their membranes. In each disease subtype, these cells, and therefore the receptors, are different and are known by different “names”. For example, the receptors on the cells of acute lymphoblastic leukaemia or some B-type non-Hodgkin’s lymphomas, such as large B-cell non-Hodgkin’s lymphoma, are called CD19; those of Hodgkin’s lymphoma or some types of T-type non-Hodgkin’s lymphoma, CD30; or those of multiple myeloma, CD38. These “DCs” (cluster of differentiation) are cellular antigens that are expressed on the membranes of tumour cells and are made up of amino acid chains.
But, back to the T-lymphocytes. As mentioned above, they are “trained” to generate an immune response: to kill foreign cells in the body. These T-lymphocytes eradicate many foreign cells that are encountered, yet not all of them and not specifically. A CART could be defined as “a calibration of these cells”. It would like taking these T-cells that the patient already has and which know how to destroy cells, and design them in the laboratory specifically to kill the specific receptors on the tumour cells: CD19, CD30, CD38…. depending on the disease being treated.
When a CART is performed, T-lymphocytes are first extracted from the patient to be treated. Using a virus with a very long incubation period such as a lentvirus, immunologists will infect these T-lymphocytes with a perpetual anti-CD19 virus, for example in the case of B-lymphoblastic leukaemias and some types of B lymphomas, and multiply them in the laboratory, specifically in a clean room. A clean room is a room specially designed to obtain low levels of contamination. It must have the strictly controlled environmental parameters: airborne particles, temperature, humidity, air flow, internal air pressure, lighting…
Subsequently, after a conditioning cycle with chemotherapy, these anti-CD19 cells will be infused back into the patient. In this way, the patient will be left with perpetual T-lymphocytes loaded with “anti-CD19 weaponry” called anti-CD19 CARTs.
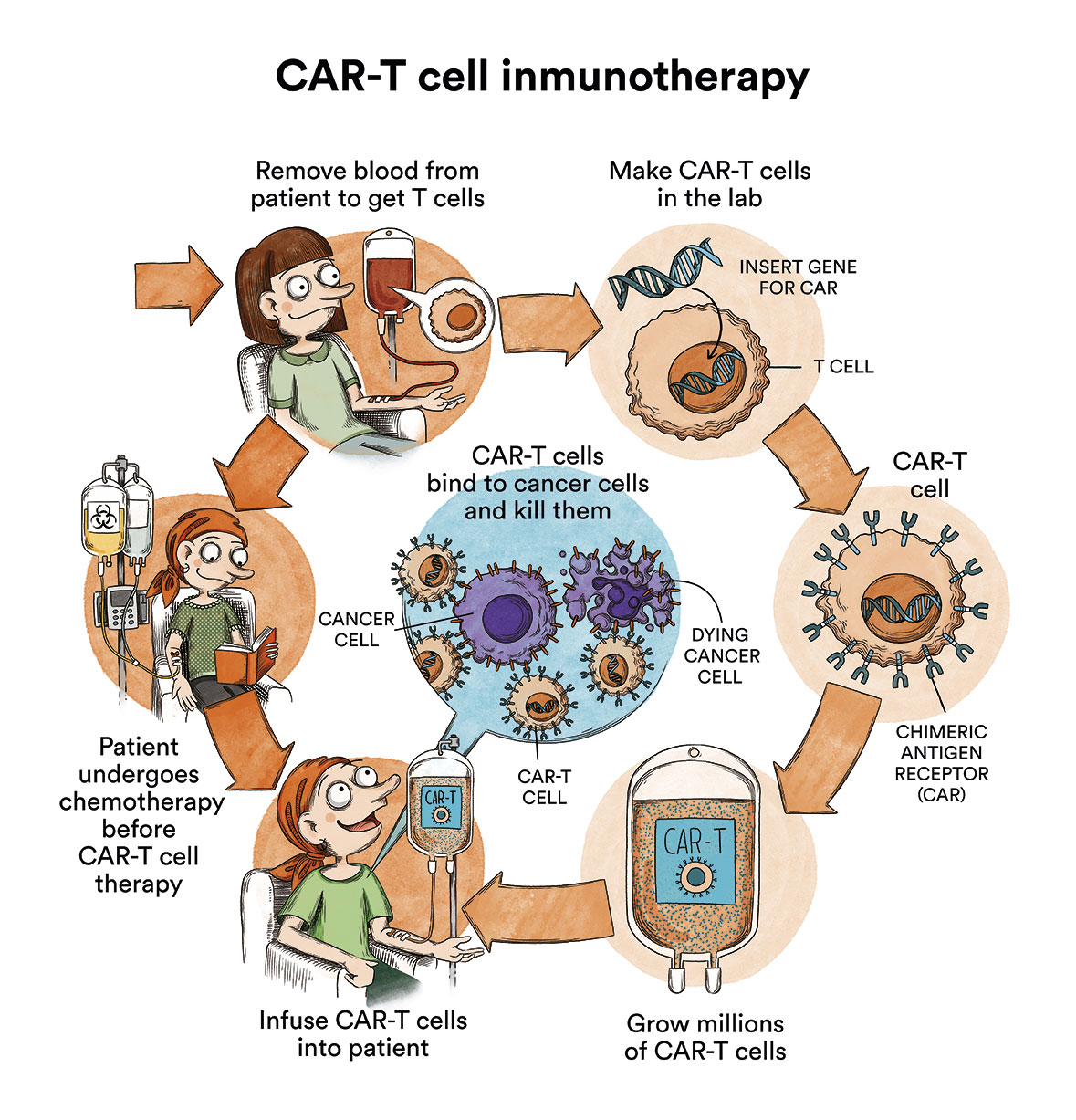
If you want to know more about CAR-T immunotherapy, how it was created and in which hospitals in Spain it is available, please see “CART immunotherapy or how to re-engineer your own cells to attack cancer (content in spanish)” on the Unstoppable Against Leukaemia blog. If you want to find out more information of CAR-T cells in English please read: CAR T Cells: Engineering Patients’ Immune Cells to Treat Their Cancers (National Cancer Institute).
Juanxo, 25 years old
Acute lymphoblastic leukaemia.
“I am here, and I remain unstoppable thanks to research. I was diagnosed with leukaemia when I was 19 and underwent a bone marrow transplant from an anonymous donor located by the Foundation. But I had an early relapse. When things were looking worse and doctors were less optimistic, a small thread of hope appeared in the form of a clinical trial: a CAR-T immunotherapy. As they taught me, you must try until the last bullet is exhausted and that is how today I am completely cured.”
Check-point inhibitors
Our body’s immune system is a highly controlled system. It has to act, yet in a regulated manner, if it were to become out of control, it can lead to situations of “collapse”. For example, when faced with an infection, if our immune system responds very poorly, this will be a major problem for fighting the infection and it will not succeed. But if the immune system “overdoes it”, a brutal inflammation occurs in our body. This very severe clinical manifestation is called “cytokine storm (content in spanish)” or “cytokine release syndrome”. This is why there are immune control points in our bodies called check-points.
When the immune response is about to occur, and proteins on the surface of T cells bind to other proteins on, for example, a healthy cell, immune checkpoints are triggered and send an “off” signal to the T cells to prevent them from attacking the healthy cell. But if it occurs when the T-lymphocyte goes to attack a tumour cell, this can prevent the immune system from destroying the cancer.
Drugs called check-point inhibitors block check-point proteins from binding to other proteins. This prevents the “off” signal from being sent, to allow the T cells to destroy the cancer cells. It would be a “lifting of barriers” to help the immune system fight cancer. For example, some types of lymphoma use drugs such as pembrolizumab or nivolumab, which target the PD-1 protein, inhibiting it by allowing T cells to act against cancer cells.
– Check-point inhibitors. National Cancer Institute
What are the side effects of immunotherapies?
Indeed, helping the immune system to better fight cancer comes at a price and yes, there are side effects of these treatments that vary depending on the type of treatment. One of the common side effects is cytokine release syndrome (content in spanish), which is a systemic (whole body) inflammatory response that can range from persistent fever and mild symptoms of headache or rash to severe hypotension, confusion, liver and kidney impairment, hence close monitoring of side effects is essential, especially at the start of treatment by professional specialists.
- New drugs, new side effects: complications of cancer immunotherapy. National Cancer Institute
Links of interest on other topics related to immunotherapies
Immunotherapy. American Cancer Society
Immunotherapy to Treat Cancer. National Cancer Institute
Immunotherapy. Cancer Research UK
* In accordance with Law 34/2002 on Information Society Services and Electronic Commerce (LSSICE), the Josep Carreras Leukemia Foundation informs that all medical information available on www.fcarreras.org has been reviewed and accredited by Dr. Enric Carreras Pons, Member No. 9438, Barcelona, Doctor in Medicine and Surgery, Specialist in Internal Medicine, Specialist in Hematology and Hemotherapy and Senior Consultant of the Foundation; and by Dr. Rocío Parody Porras, Member No. 35205, Barcelona, Doctor in Medicine and Surgery, Specialist in Hematology and Hemotherapy and attached to the Medical Directorate of the Registry of Bone Marrow Donors (REDMO) of the Foundation).
Become a member of the cure for leukaemia!


