Hodgkin’s lymphoma in children and teenagers
The information provided on www.fcarreras.org is intended to support, not replace, the relationship that exists between patients/visitors to this website and their physician.
Victoria
Hodgkin's lymphoma.
“I’m Victoria. When I was 11 years old I was diagnosed with Hodgkin’s lymphoma. The treatment was hard, but I was always smiling. I wanted to tell you that you can get out of this, that I have won the battle and that I am doing well.”
Information endorsed by… ![]()
Information reviewed by Dr. Rocío Parody Porras, Doctor specialising in Haematology. Member of the Management of REDMO (Bone Marrow Donor Registry) and of the Foundation’s medical team. Barcelona Medical Association (Co. 35205)
What is Hodgkin’s lymphoma in children and teenagers and who does it affect?
Hodgkin’s lymphoma is most common in children and adolescents aged between 15 and 19 years old. Treatment for children and adolescents is different to treatment for adults. See adult Hodgkin’s lymphoma
Unlike non-Hodgkin’s lymphomas, of which there are more than 60 subtypes of completely different clinical presentation and course, the latest World Health Organisation classification divides Hodgkin’s lymphoma into two main subtypes: classical Hodgkin’s lymphoma and nodular lymphocyte-predominant Hodgkin’s lymphoma.
- Classical Hodgkin’s lymphoma
This is the most common type of Hodgkin’s lymphoma and it constitutes 95% of all Hodgkin’s lymphomas. It is most frequent in adolescents. When a sample of lymph node tissue is analysed under a microscope, the Hodgkin’s lymphoma cells are observed, known as Reed-Sternberg cells. These cells are actually large and abnormal lymphocytes that occasionally contain more than one cell nucleus. These characteristic cells express two membrane antigens: CD30 and CD15. The expression of CD30 is important in terms of considering the treatment of the disease with specific monoclonal antibodies.
Classical Hodgkin’s lymphoma can be divided into 4 different subtypes and, therefore, each of these can be divided into 4 different stages. First we will take a look at the different subtypes:
- Hodgkin’s lymphoma with nodular sclerosis. This is the most common subtype (80% of diagnoses of classical Hodgkin’s lymphoma) and is most frequent in older children, adolescents and young adults. It is common for this disease to affect mediastinum lymph nodes (centre of the chest) in these patients. A chest mass is therefore often present upon diagnosis.
- Mixed cellularity Hodgkin’s lymphoma. This subtype tends to affect the youngest children, often under 10 years old. It is associated with a history of Epstein-Barr virus infection (EBV, also known as human herpes virus 4) and is frequently found in the lymph nodes of the neck upon diagnosis.
- Classical lymphocyte-rich Hodgkin’s lymphoma.. This subtype is rare in children. When a sample of lymph node tissue is viewed under the microscope, Reed-Sternberg cells, many normal lymphocytes and other blood cells are found.
- Lymphocyte-depleted Hodgkin’s lymphoma. This is another rare subtype in children and occurs more frequently in adults and/or adults with human immunodeficiency viruses (HIV). When a sample of lymph node tissue is viewed under the microscope, many large, bizarrely shaped cancer cells, few normal lymphocytes and other blood cells can be observed.
- Nodular lymphocyte-predominant Hodgkin’s lymphoma
This type of Hodgkin’s lymphoma is rare (less than 5% of all Hodgkin’s lymphoma diagnoses). In childhood it occurs in older children. When looking at the affected lymphoid tissue under a microscope, the cancer cells have a very distinctive “popcorn” shape. These cells do not express the CD30 nor CD15 antigens, but they do express CD20. This type of lymphoma is often diagnosed following a swollen lymph node in the neck, armpit or groin. This type of Hodgkin’s lymphoma has a very different prognosis and evolution to classical Hodgkin’s lymphoma.
As mentioned previously, Hodgkin’s lymphoma is also classified into stages that enable haematologists to know the extent or involvement of the cancerous cells. In other words, it is necessary to know how much disease is present in the patient upon diagnosis. It is an important diagnostic tool as it then facilitates treatment selection and intensity. This is called Ann Arbor staging.
- Stage 1: the lymphoma affects one single area or organ outside the lymphatic system.
- Stage 2: two or more lymph node regions on the same side of the diaphragm are affected.
- Stage 3: several lymph node regions on both sides of the diaphragm are affected.
- Stage 4: one or more extranodal regions (bone marrow, liver, lungs) are affected.
The stage corresponding to the Ann Arbor staging system is accompanied by the letters A, B, E or S:
- No B symptoms present at diagnosis (fever, significant weight loss, night sweats).
- B symptoms present.
- An extranodal region is affected as a result of contiguity due to the involvement of a lymph node or cluster of nodes.
- The spleen is affected.
All these classifications make it possible to elucidate the risk group of the diagnosed lymphoma and thus propose the most appropriate treatment.
What are the causes of Hodgkin’s lymphoma?
 Today, the causes of Hodgkin’s lymphoma are unknown. In principle, it is not an infectious or hereditary disease, there are no environmental factors that are clearly related to its development and, despite the fact that the Epstein-Barr virus can be detected in some cases, the casual relationship between the virus and the development of the disease is not well understood at this time, however numerous research groups are working to elucidate this relationship.
Today, the causes of Hodgkin’s lymphoma are unknown. In principle, it is not an infectious or hereditary disease, there are no environmental factors that are clearly related to its development and, despite the fact that the Epstein-Barr virus can be detected in some cases, the casual relationship between the virus and the development of the disease is not well understood at this time, however numerous research groups are working to elucidate this relationship.
According to the United StatesNational Cancer Institute, “it is possible that the Epstein-Barr virus infection and family history of Hodgkin’s lymphoma increase the risk of childhood Hodgkin’s lymphoma”. These are known as “risk factors”, but do not in any way mean that a person will develop cancer.
The following are risk factors for Hodgkin’s lymphoma:
- Being infected with the Epstein-Barr virus (EBV).
- Having a personal history of mononucleosis.
- Being infected with human immunodeficiency viruses (HIV).
- Having certain immune system diseases, such as autoimmune lymphoproliferative syndrome.
- Having a weakened immune system following an organ transplant or due to medication given to prevent the body from rejecting the organ.
- Having a parent or sibling with a history of Hodgkin’s lymphoma.
What are the symptoms of Hodgkin’s lymphoma?
In most cases, patients have no symptoms present upon diagnosis (60-70% of cases). The most frequent reason for a consultation is the appearance of an enlarged lymph node. These lymph nodes are preferentially located in the cervical region (they are often mistaken for swollen lymph nodes secondary to oral, dental or ear infections). Sometimes these enlarged nodes are located in the armpit or inguinal region. Patients often attend a consultation due to a persistent dry cough and a chest X-ray shows a mass in the mediastinum (the area between the two lungs). Less frequently, the patient presents with abdominal pain as a result of an enlarged spleen. Patients only present with so-called B symptoms in 25-30% of cases (weight loss of more than 10% in the last six months with no known cause, evening fever and night sweats). Occasionally, patients may present with generalised skin itching and lesions due to scratching lasting for several months prior to diagnosis of the disease (10-15% of patients).
Some of these symptoms can include:
Painless swollen lymph nodes near the collarbone, in the neck, chest, armpits or groin.
- Unexplained fever.
- Unexplained weight loss/loss of appetite
- Excessive night sweats.
- Severe fatigue.
- Itchy skin.
- Cough.
- Difficulty breathing, especially when lying down.
Fever, unexplained weight loss and excessive night sweats are known as B symptoms. Whether or not they are present at diagnosis is important in order to classify the lymphoma.
How is Hodgkin’s lymphoma diagnosed?
Hodgkin’s lymphoma should be diagnosed as follows:
- a physical examination and medical history
- blood test (haemogram and biochemistry)
- imaging test to detect the presence of pathological lymph nodes or other affected areas (at present, the test is positron emission tomography associated with computed tomography – PET/CT) and, in cases where PET/CT is negative, a bone marrow biopsy is no longer recommended.
- biopsy of a suspicious lymph node. Aspiration of a swollen lymph node, which is a very frequently used strategy for the diagnosis of solid tumours in medical oncology. It is not a valid technique for the diagnosis of lymphomas in general as it does not allow the structure of the lymph node to be seen under the microscope. In some cases, core needle biopsy can be used.
What is the treatment for Hodgkin’s lymphoma?
 Before the most appropriate treatment can be considered, the diagnosis must be accurate and the child or adolescent must be treated by a medical team with prior experience in childhood cancer.
Before the most appropriate treatment can be considered, the diagnosis must be accurate and the child or adolescent must be treated by a medical team with prior experience in childhood cancer.
Hodgkin’s lymphoma is the most common haematological cancer that can be cured to date. In recent years, we have significantly improved long-term survival rates for patients, especially in the youngest patients. In general, the treatment of Hodgkin’s lymphoma is based on the use of polychemotherapy, sometimes associated with radiotherapy to the affected area, high-dose chemotherapy with autologous transplant of haematopoietic progenitors and, today, we also have so-called “new drugs” for the treatment of this disease.

First line treatment
The standard first-line chemotherapy regimen is called ABVD (based on the combination of the following drugs: adriamycin, bleomycin, vinblastine and dacarbazine). ABVD chemotherapy is administered intravenously on an outpatient basis for most patients. The cycle is repeated every 28 days with two administrations, the first on day 1 and the second on day 14. In patients diagnosed in early stages without poor prognostic factors, the recommended treatment is the administration of two cycles of ABVD and complementary radiotherapy to the affected areas (20Gys). In patients diagnosed at early stages but with poor prognostic factors, four cycles of AVBD and complementary radiotherapy (30 Gys) are recommended. Treatment of advanced stage patients involves six cycles of ABVD. If the PET/CT scan following chemotherapy is negative, complementary radiotherapy treatment is not generally necessary. There are other more intensive chemotherapy regimens which, although more effective in controlling the disease, are also more toxic, and are not widely used in our country (e.g. escalated BEACPP).
Second-line and subsequent treatment.
Although the results of first-line treatment are very good, 5-15% of patients are primarily refractory (do not respond to first-line treatment) and 30% of patients who, following achieving complete remission, subsequently relapse. These patients are treated with second line chemotherapy regimes. These treatments are more intense than first-line treatments and, above all, use different drugs to those included in first-line treatment. If the patient achieves a response, consolidation with high-dose chemotherapy and autologous haematopoietic stem cell transplant is indicated.. Patients who relapse following autologous transplant have a worse prognosis. Until relatively recently, patients who relapsed following an autologous transplant could be candidates for an allogeneic progenitor haematopoietic stem cell transplant from an identical sibling, a matched unrelated donor and, more recently, from a haploidentical family donor.
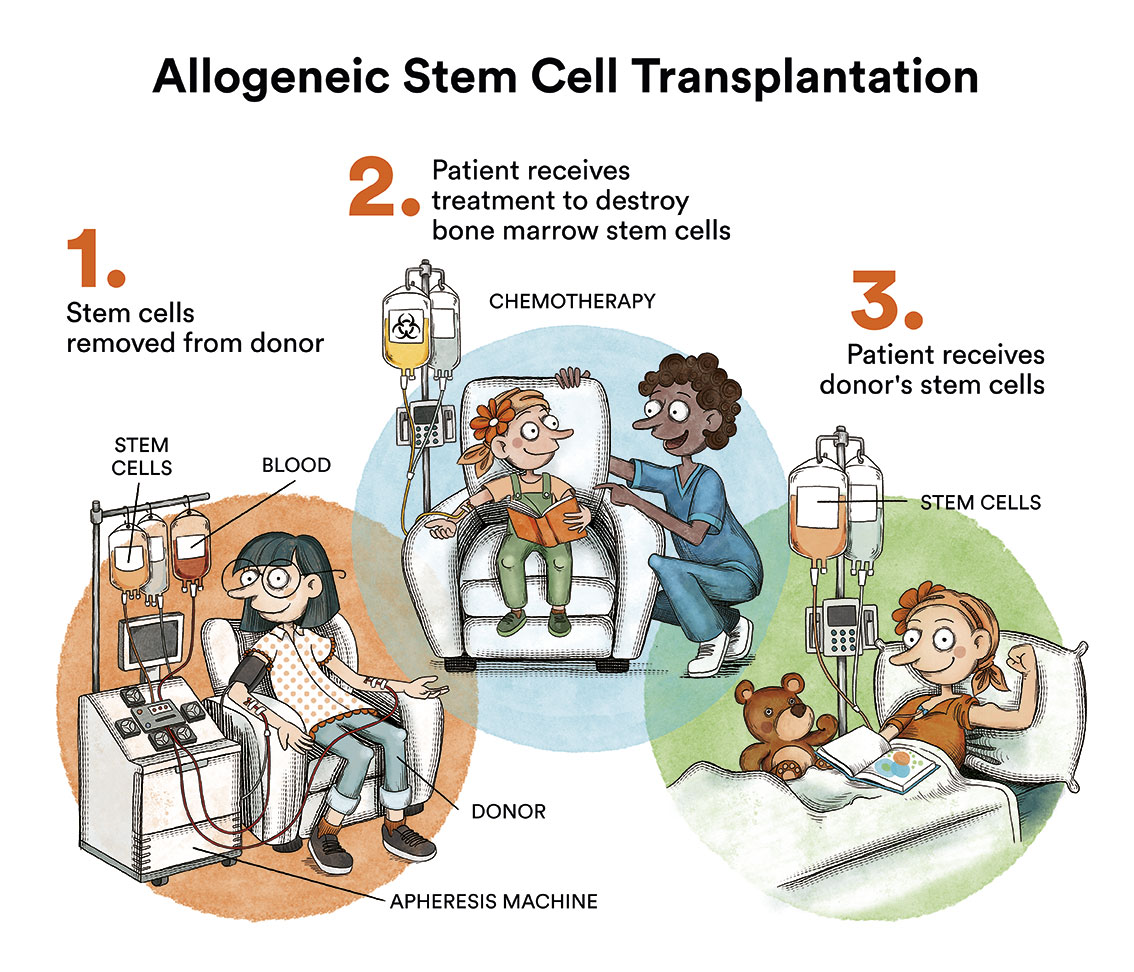
We now have access to “new drugs” or targeted therapies for treating Hodgkin’s lymphoma, which are currently indicated for patients who have relapsed or are refractory to previous treatment.
- Monoclonal antibodies:
Monoclonal antibodies are immune system proteins that are created in a laboratory. These antibodies attach to specific targets on cancer cells or to other cells that facilitate the formation of malignant cells. These monoclonal antibodies destroy cancer cells, block them or prevent them from disseminating. These antibodies are infused into the patient. This means that they are grafted into the bloodstream by way of a catheter.
One such monoclonal antibody used for relapsed or refractory Hodgkin’s lymphoma patients is brentuximab vedotin, an anti-CD-30 monoclonal antibody that has proven to be very effective and to have a very acceptable toxicity profile in relapsed or refractory patients following autologous transplant, or in patients who have failed two different lines of prior chemotherapy and are not candidates for autologous haematopoietic stem cell transplant.
- Checkpointinhibitors:
The second group of new drugs we are currently using to treat these patients are checkpoint inhibitors (immune checkpoint inhibitors) which are currently indicated in patients who relapse following autologous transplant and who also fail treatment with brentuximab vedotin. Nivolumab and pembrolizumab are very effective checkpoint inhibitors that are well tolerated by most patients. Checkpoint inhibitors are drugs that help the body’s own cells attack cancer cells. Checkpoint inhibitors are a type of immunotherapy (therapies that use the body’s own immune system to fight cancer).
- Some treatments are standard and others are being tested in clinical trials. A treatment clinical trial is a research study undertaken in order to improve current treatments or obtain information regarding new treatments for cancer patients. A number of clinical trials of anti-CD-30 CAR-T immunotherapies (a protein expressed by the cancer cells of many types of Hodgkin’s lymphoma) are currently under way around the world for patients who have exhausted all other therapeutic options available in the healthcare pipeline.
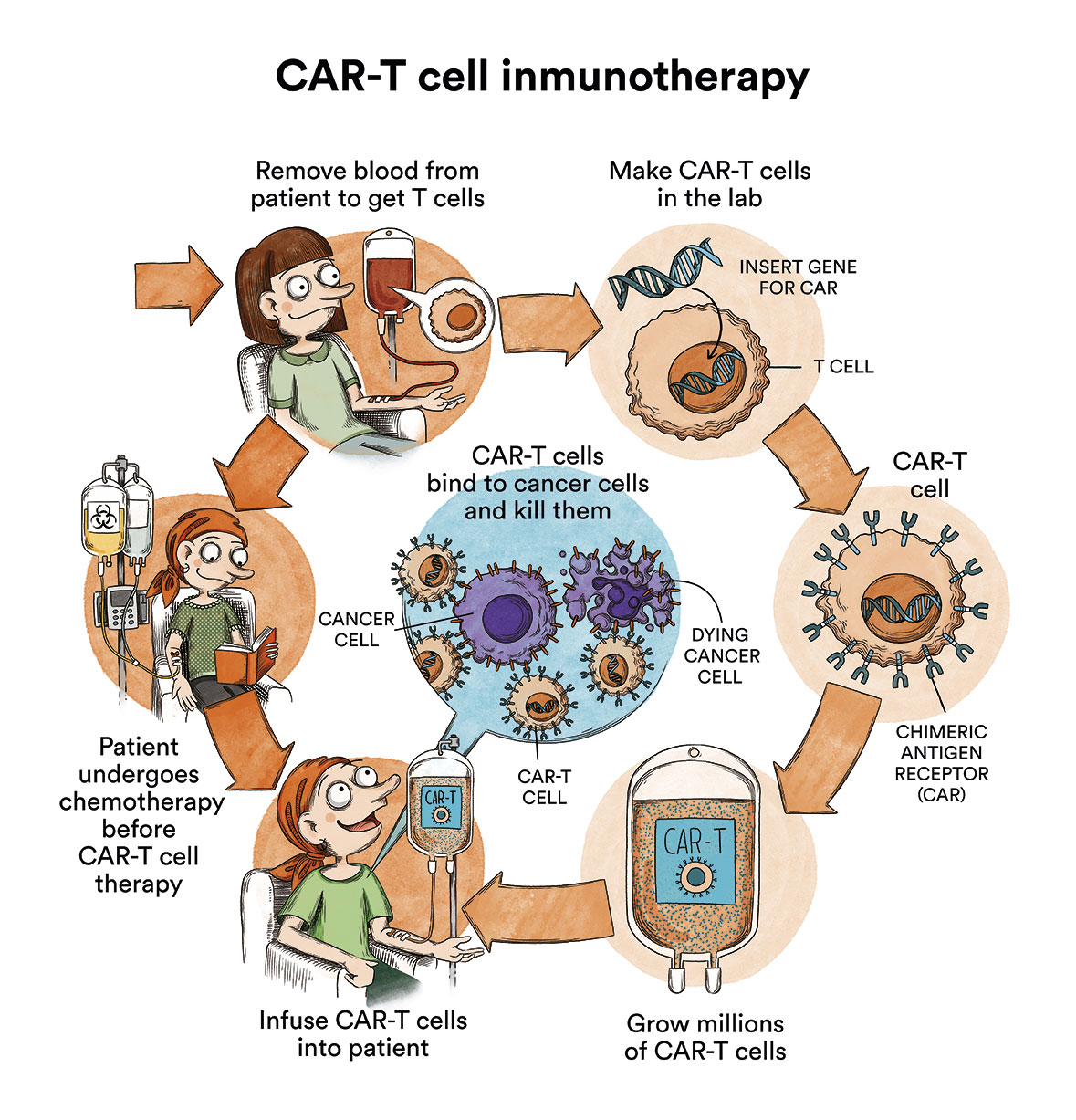
What are the chances of children and teenagers with Hodgkin’s lymphoma being cured?
As with any neoplastic disease, the prognosis depends on the histological type of Hodgkin’s lymphoma, its extension and response to treatment. In the absence of adverse factors, and where they have been treated correctly, the possibility of these patients being cured is greater than 80%.
Despite the good general prognosis of patients with Hodgkin’s lymphoma, the prognosis is significantly less favourable in patients who do not achieve complete remission following the initial treatment, or patients with late relapse.
Links of interest concerning medical issues relating to Hodgkin’s lymphoma in children and teenagers
Childhood Hodgkin Lymphoma Treatment. National Cancer Institute
Treating Hodgkin Lymphoma in Children. American Cancer Society
Hodgkin Lymphoma in children. Children with Cancer UK
Useful links: local entities (resources and services)
All these organisations are external to the Josep Carreras Foundation.
ANDALUCÍA
ARAGÓN
ASTURIAS
CASTILLA LA MANCHA
CASTILLA LEÓN
CATALUÑA
VALENCIAN COMMUNITY
EXTREMADURA
GALICIA
BALEARIC ISLANDS
CANARY ISLANDS
LA RIOJA
MADRID
- AAA (asociación de adolescentes y Adultos Jóvenes con Cáncer)
- ASION
- FUNDACIÓN CAICO
- FUNDACIÓN ALADINA
- FUNDACIÓN UNOENTRECIENMIL
MURCIA
NAVARRA
BASQUE COUNTRY
Support and assistance
We also invite you to follow us through our main social media (Facebook, Twitter and Instagram) where we often share testimonies of overcoming this disease.
If you live in Spain, you can also contact us by sending an e-mail to imparables@fcarreras.es so that we can help you get in touch with other people who have overcome this disease.
* In accordance with Law 34/2002 on Information Society Services and Electronic Commerce (LSSICE), the Josep Carreras Leukemia Foundation informs that all medical information available on www.fcarreras.org has been reviewed and accredited by Dr. Enric Carreras Pons, Member No. 9438, Barcelona, Doctor in Medicine and Surgery, Specialist in Internal Medicine, Specialist in Hematology and Hemotherapy and Senior Consultant of the Foundation; and by Dr. Rocío Parody Porras, Member No. 35205, Barcelona, Doctor in Medicine and Surgery, Specialist in Hematology and Hemotherapy and attached to the Medical Directorate of the Registry of Bone Marrow Donors (REDMO) of the Foundation).


