Acute myeloid leukaemia in adults
Acute myeloid leukaemia is the most prevalent type of leukaemia in the adult population. Leukaemia affects more than 6,000 people in Spain every year.
The information provided on www.fcarreras.org is intended to support, not replace, the relationship that exists between patients/visitors to this website and their physician.
Information provided by Dr. Irene Garcia Cadenas. Specialist in Haematology at the Hospital Sant Pau in Barcelona. Barcelona Medical Association (Co. 42612).

Álvaro, 52 years old
Acute Myeloid Leukaemia.
“During the entire healing process of my leukemia, I only had one totem: Here and now. I invite you to share my experience reading my Ebook (in Spanish)”
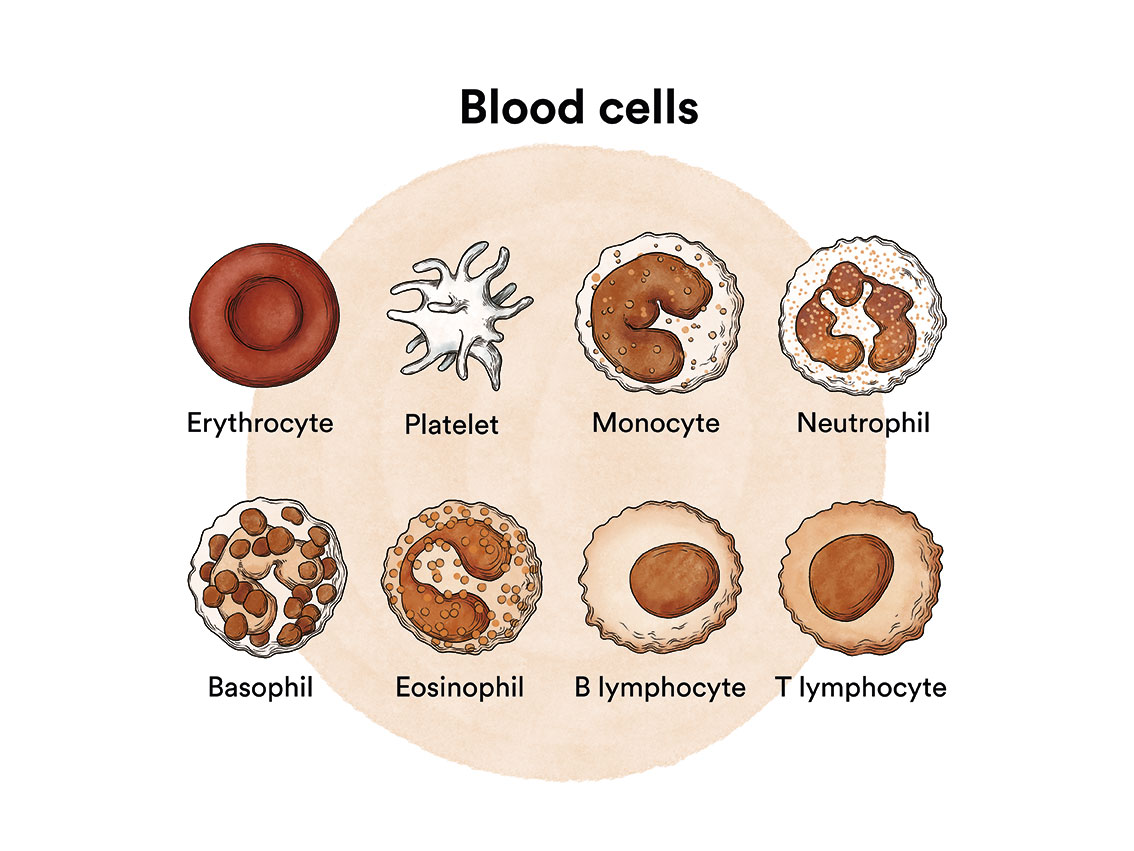
What is leukaemia, bone marrow and what are the types of blood cells?
Leukaemia is a type of blood cell cancer that originates in the bone marrow. See section Leukaemia, bone marrow and blood cells.
As we have seen in ‘Leukaemia, bone marrow and blood cells’, the bone marrow makes blood stem cells (immature cells) that will eventually develop into mature blood cells. A blood stem cell becomes a myeloid stem cell or a lymphoid stem cell.
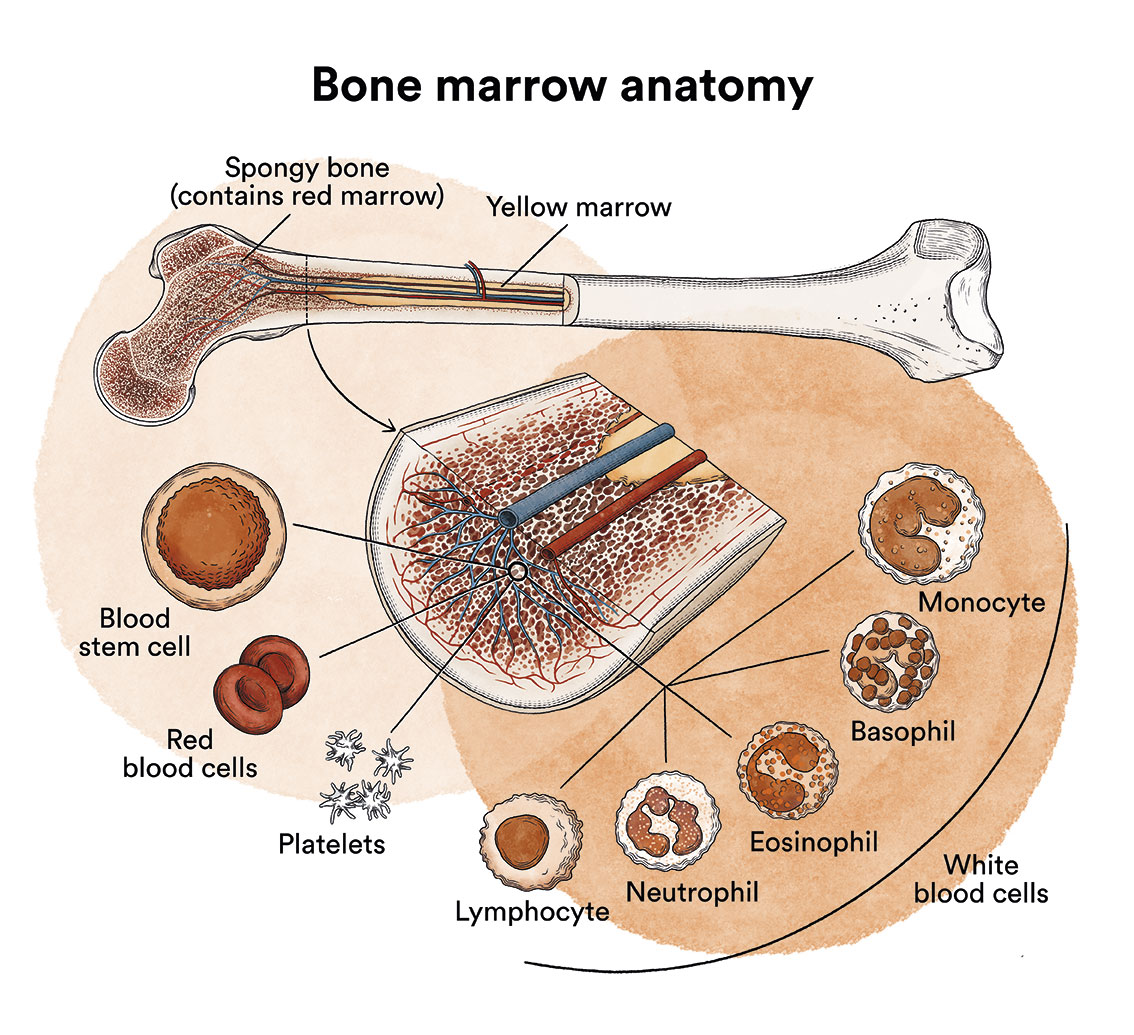
A myeloid stem cell develops into one of the three types of mature blood cells:
- Red blood cells, which carry oxygen to other tissues and organs in the body.
- Granulocytes, white blood cells that help fight infection and other diseases.
- Platelets, which help blood to clot and prevent bleeding.
Acute leukaemias are a group of neoplastic diseases characterised by malignant transformation and uncontrolled production of immature haematopoietic cells. There are two main types depending on the cell line affected: acute lymphoblastic leukaemia (ALL) and acute myeloblastic or myeloid leukaemia (AML).
What is adult acute myeloid leukaemia and who does it affect?
Acute myeloid leukaemia (also known as acute myeloblastic leukaemia, acute myelogenous leukaemia, acute granulocytic leukaemia or AML) is the most common type of acute leukaemia in adults, although it can sometimes be diagnosed in children. The median age of onset is 64 years and most patients are in the 60-75 age range. This type of leukaemia accounts for 40% of all leukaemias in the western world. Its incidence in Spain is estimated to be around 15 new cases per million inhabitants per year.
 Acute myeloid leukaemia is sometimes the end stage of other diseases such as myelodysplastic syndromes or chronic myeloproliferative syndromes. Similarly, acute myeloid leukaemia can occur years after chemotherapy and/or radiotherapy for the treatment of another malignancy; these acute myeloid leukaemias have classically been referred to as secondary leukaemias.
Acute myeloid leukaemia is sometimes the end stage of other diseases such as myelodysplastic syndromes or chronic myeloproliferative syndromes. Similarly, acute myeloid leukaemia can occur years after chemotherapy and/or radiotherapy for the treatment of another malignancy; these acute myeloid leukaemias have classically been referred to as secondary leukaemias.
Another peculiarity is that its incidence is higher among people with certain chromosomal disorders such as Down syndrome or Fanconi anaemia.
What are the causes of acute myeloid leukaemia in adults?
The specific causes of most cases of adult acute myeloid leukaemia are not known. However, there are some risk factors that are associated with a higher likelihood of developing an acute myeloid leukaemia. A risk factor is anything that increases the likelihood that a person will develop cancer.
Risk factors associated with acute myeloid leukaemia are, according to the American Society of Clinical Oncology:
- Age. Acute myeloid leukaemia is more common in older adults, but occurs in all ages.
- Genetic disorders. Acute myeloid leukaemia occurs most commonly in people with the following inherited disorders:
- Down Syndrome
- Ataxia-telangiectasia
- Li-Fraumeni syndrome
- Klinefelter’s syndrome
- Fanconi anaemia
- Wiskott-Aldrich syndrome
- Bloom syndrome
- Familial platelet disorder syndrome
- Germline mutations that are present at birth; the most frequent are changes in the GATA2, ETV6, CEBPA and RUNX1 genes.
- High radiation doses. People who have been exposed to high levels of radiation may be more likely to develop acute myeloid leukaemia. This includes people who have received radiotherapy for another cancer or long-term survivors of atomic bomb or radioactive leak accidents. Electromagnetic fields generated by high-voltage power lines have not been shown to cause acute myeloid leukaemia. Mobile phone use is not a known risk factor for acute myeloid leukaemia.
- Chemotherapy. People who have received chemotherapy for another cancer may develop acute myeloid leukaemia related to previous therapy.
- Chemicals. Prolonged contact with benzene-containing products (found in petroleum, cigarette smoke and some industrial workplaces) increases the risk of acute myeloid leukaemia. However, exposure to industrial solvents and hair dyes has not been shown to increase a person’s risk of developing leukaemia.
- Other bone marrow disorders. People with other bone marrow disorders, including myeloproliferative disorders, may develop acute myeloid leukaemia over time. “Myelo” means from the bone marrow and “proliferative” means multiplying abundantly. These conditions include: Chronic myeloid leukaemia, polycythaemia vera, myelofibrosis and essential thrombocytosis. There is also an increased risk of progression to AML in people with myelodysplastic syndromes or aplastic anaemia.
Leukaemia, like other cancers, is not contagious. See section Leukaemia, bone marrow and blood cells.
How is acute myeloid leukaemia classified?
In the 1970s a group of French, American and British experts defined the FAB classification, differentiating AML into subtypes, from M0 to M7, according to the type of cell from which the leukaemia develops and how mature the cells are. This was mainly based on the appearance of the leukaemic cells under the microscope after routine staining and on the expression of certain proteins in the leukaemic cell (immunophenotyping).
| FAB Subtype | Name | Frequency |
|---|---|---|
| AML M0 | Undifferentiated acute myeloblastic leukemia | 2-5% |
| AML M1 | Acute myeloblastic leukemia with minimal maturation | 15-20% |
| AML M2 | Acute myeloblastic leukemia with maturation | 25-30% |
| AML M3 | Acute promyelocytic leukemia (APL) | 10-15% |
| AML M4 | Acute myelomonocytic leukemia | 15-30% |
| AML M5 | Acute monocytic leukemia 10-15% | 10-15% |
| AML M6 | Acute erythroid leukemia 3-4% | 3-4% |
| AML M7 | Acute megakaryoblastic leukemia 1% | 1% |
However, there are now two new, recently updated classifications (WHO 2022 and ICC 2022) that focus on cytogenetic and molecular alterations in AML, which are highly related to the prognosis, and may be susceptible to some targeted drug therapy.
The most commoncytogenetic alterations in AML are translocations; displacement of a fragment from one chromosome to another chromosome (indicated as t). Example: t(8;21), a fragment of chromosome 8 moves to an area of chromosome 21; or within the same chromosome, t(16;16). Cytogenetic inversions can also be observed, which is when a chromosome segment changes direction within the chromosome itself (indicated as inv).
Translocations or inversions detected in cytogenetic studies result in rearrangements of genes located in the affected chromosomal regions. These are represented by the names of the genes involved, so in the case of t(8;21) it will generate a rearrangement of the RUNX1 and RUNX1T1 genes. Cytogenetic alterations have proven to be a very important prognostic factor and are used in most treatment protocols to determine their intensity.
In recent years, mutations in one or more genes have been described in the leukaemic cells of most patients. Some of them have been shown to have prognostic significance and to be relevant in defining the intensity of treatment.
Therefore, nowadays, both chromosomal and molecular alterations are assessed in order to establish treatment protocols.
Knowing the subtype of acute myeloid leukaemia is very important, as it affects both a patient’s prognosis and the choice of the best treatment. For example, the acute promyelocytic leukaemia subtype (AML3) is often treated with drugs that are different from those used for other subtypes of acute myeloid leukaemia.
Depending on the chromosomal abnormalities (cytogenetic) some subtypes of acute myeloid leukaemia can be established with a more or less favourable prognosis. These prognostic factors help haematologists determine the risk of a person’s leukaemia returning after treatment, and therefore whether they should receive more or less intensive treatment. Some of these criteria are:
- Favourable anomalies:
- Translocation between chromosomes 8 and 21 (most commonly seen in patients with AML2)
- Translocation or inversion of chromosome 16
- Translocation between chromosomes 15 and 17 (most commonly seen in patients with AML3 – acute promyelocytic leukaemia)
- Unfavourable anomalies:
- Deletion (loss) of part of chromosome 5 or 7
- Translocation or inversion of chromosome 3
- Translocation between chromosomes 6 and 9
- Translocation between chromosomes 9 and 22
- Chromosome 11 abnormalities (at the q23 locus)
- Loss of one chromosome, thus the cell has only one copy instead of the normal two (monosomy)
- Complex changes (involving 3 or more chromosomes)
Any molecular changes at diagnosis are also very important for understanding prognosis and treatment options along with chromosomal changes. The most common molecular genetic changes associated with prognosis for people with AML include: NPM1, CEBP alpha, FLT3, RUNX1, ASXL1, TP53, IDH1 and IDH2.

Cristina
Acute Myeloid Leukaemia.
“10 years ago I was diagnosed with acute myeloid leukaemia. I remember with distress the time of testing, the uncertainty, not knowing what I could have. Finally, the diagnosis, treatments and a bone marrow transplant arrived. Of all my illness, what I am left with is the demonstration of love that I had from my friends and family. I felt tons of love that helped me continue fighting every day. I also appreciate the fantastic medical team I met at the two hospitals that treated me. Finally, I learned to focus on the present, that you cannot plan, that you must accept things as they come with patience and hope. To live each day and focus on that day ahead, no more, no less.”
What are the symptoms of acute myeloid leukaemia in adults?
The interval between the onset of the first symptoms and diagnosis is usually less than 3 months due to the acute nature of the disease. The symptoms of patients with acute myeloid leukaemia are the result of anaemia caused by red blood cell deficiency (feeling tired, weakness, dizziness, paleness); platelet deficiency (bruising, bleeding from the gums, nose or any other source); and granulocyte deficiency (fever and infections).
See section Leukaemia, bone marrow and blood cells.
Occasionally, patients may also experience an enlargement of the lymph nodes, liver or spleen. Specific symptoms of infiltration of the central nervous system (headache, vomiting, drowsiness, etc.), skin (disseminated nodules or areas of thickened skin), mucous membranes (inflammation of the gums), eyes (blurred vision, blindness), among others, may also be observed.
How is acute myeloid leukaemia in adults diagnosed?
In addition to basic blood and bone marrow studies (morphology, blood count, immunophenotyping) to be performed for all types of leukaemia, cytogenetic studies (to detect specific chromosomal abnormalities) and molecular studies (to detect specific genetic alterations) are essential for typing and classifying the disease. Certain cytogenetic and molecular alterations correlate with treatment sensitivity and relapse prognosis.
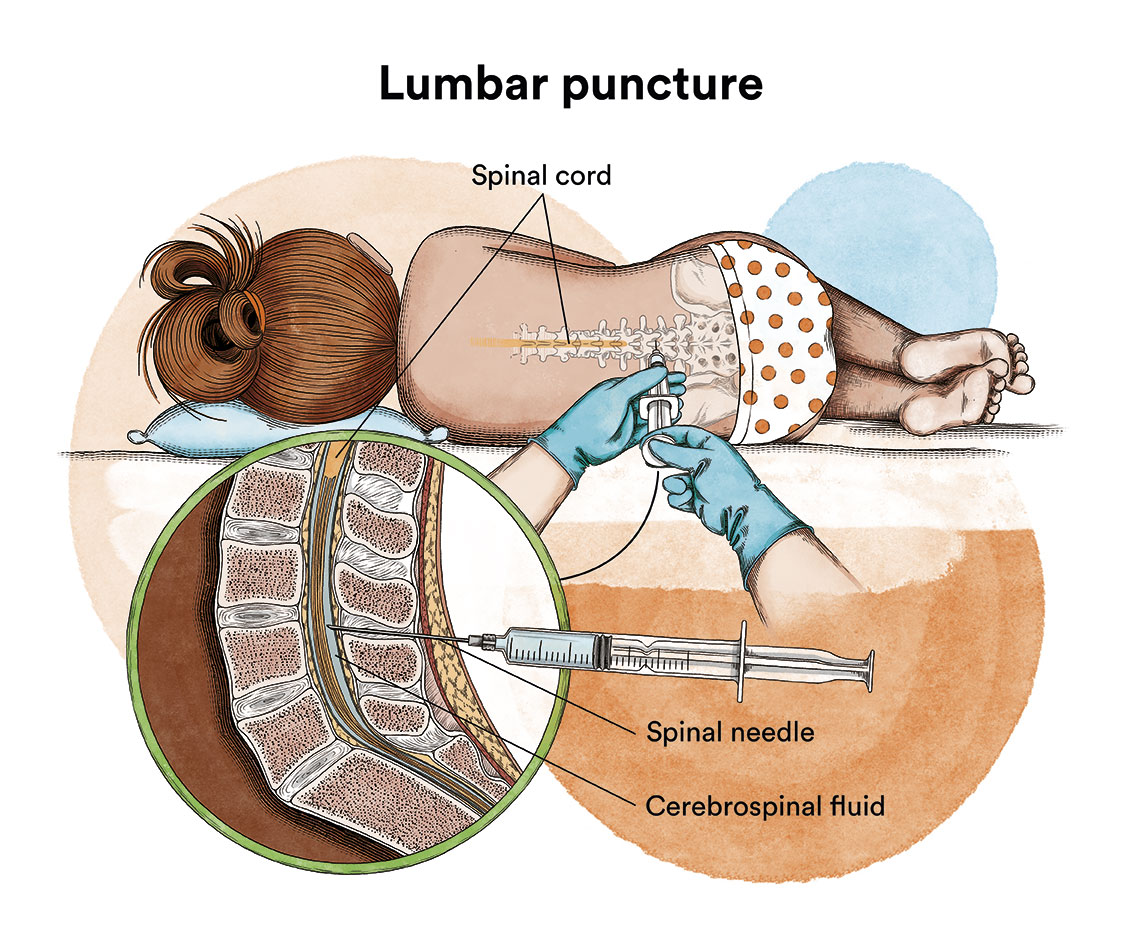
Depending on the symptoms and the subtype of leukaemia, it is sometimes necessary to check whether the disease has spread to the central nervous system by performing a lumbar puncture to analyse the fluid that surrounds the central nervous system (cerebrospinal fluid), and if it is affected, local (intrathecal) chemotherapy should be administered.
What is the treatment for acute myeloid leukaemia in adults?
Each treatment for acute myeloid leukaemia will be determined on a case-by-case basis taking into account the subtype of the disease, the patient’s age, general condition and, subsequently, the response to initial treatment.
The main goal of any treatment for leukaemia or other haematological malignancies is to achieve complete remission of the disease at a molecular level.
The most commonly used chemotherapy treatment to date in patients under 70 years of age includes 2 phases: remission induction and post-remission or consolidation. The low-dose maintenance phase of chemotherapy used for very long periods in acute lymphoblastic leukaemia (ALL) has not proven to be effective in acute myeloid leukaemia.
In general, treatment of acute myeloid leukaemia should be started as soon as possible after diagnosis, although, depending on the aggressiveness of presentation, we can wait a few days to have complete information on the main features of the disease and thus better direct treatment, provided that the patient’s general condition is good and with close clinical monitoring.
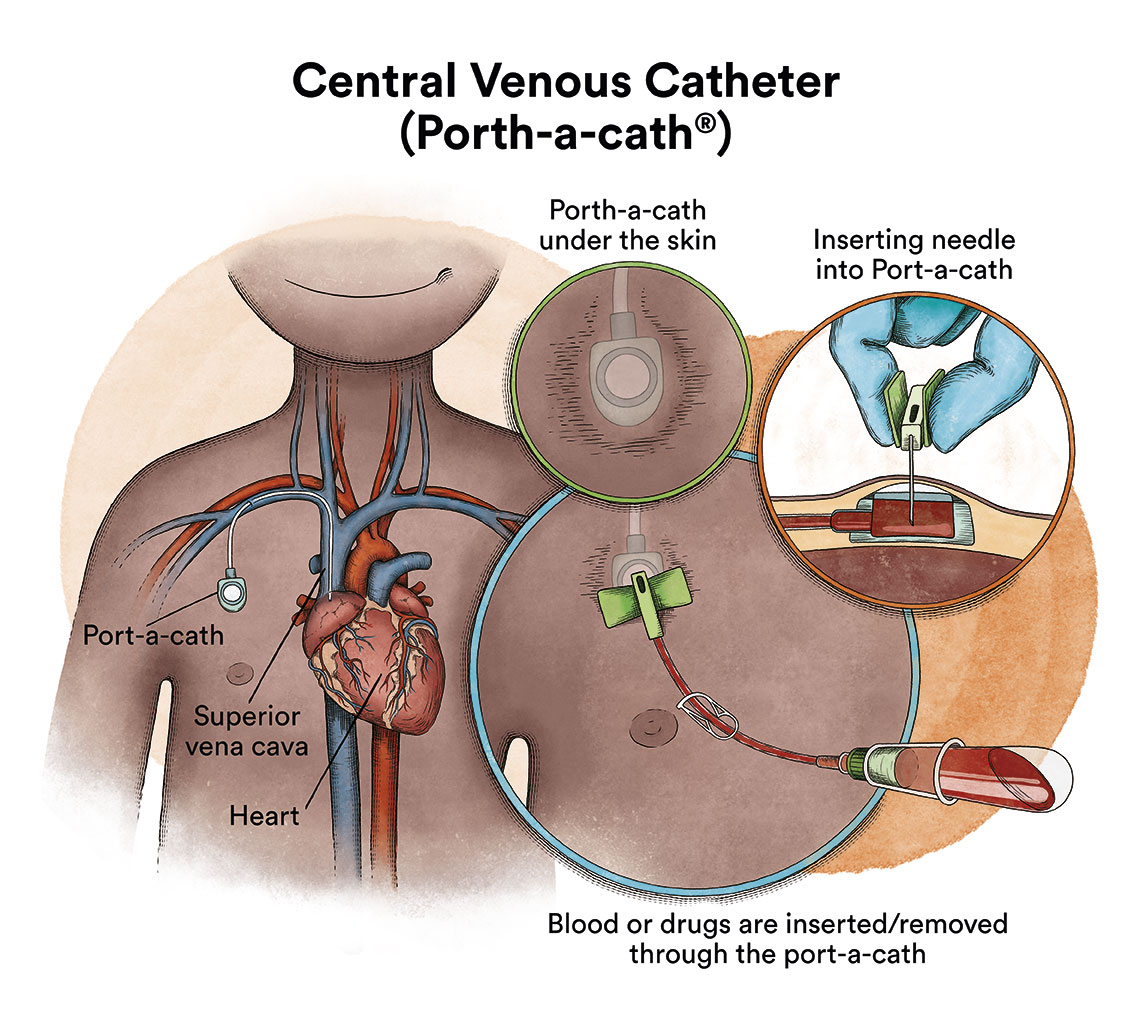
Due to the administration of chemotherapy and/or blood transfusions, in general, one of the first steps after diagnosis will be the placement of a central venous catheter to facilitate treatment, because the peripheral (arm) veins are not sufficiently resistant to endure an access through which to administer treatment and general support (serums, transfusions, chemotherapy, etc.).
1. Remission induction phase (except in acute promyelocytic leukaemia). See below)
The haematology team will consider the treatment to be used depending on the age and general condition of the patient and the presence of other diseases (known as comorbidities), as well as other aspects such as the specific genetic or chromosomal alterations of the leukaemia.
Standard induction treatment in patients without relevant comorbidities and up to 70 years of age is usually based on intensive chemotherapy including various intravenous antineoplastic agents, with the aim of eliminating leukaemic cells from the blood and bone marrow (complete cytological remission), allowing for normal production of other blood cells. A patient is considered to have achieved cytological complete remission when the number of blasts in the bone marrow is less than 5%. This clinical situation is usually achieved in 70-80% of all patients after the first treatment cycle, although it may sometimes be necessary to administer two induction cycles to achieve remission or even to change the therapeutic strategy.
 The two chemotherapy agents used are cytarabine (ara-C) and a drug from the anthracycline family, such as daunorubicin (daunomycin) or idarubicin. This regimen is often called the 7 + 3 regimen, as it consists of receiving cytarabine continuously for 7 days, along with short infusions of anthracycline on each of the first 3 days.
The two chemotherapy agents used are cytarabine (ara-C) and a drug from the anthracycline family, such as daunorubicin (daunomycin) or idarubicin. This regimen is often called the 7 + 3 regimen, as it consists of receiving cytarabine continuously for 7 days, along with short infusions of anthracycline on each of the first 3 days.
In some situations where there is a target molecule a third drug might also be added in an attempt to improve the chances of remission:
- If the FLT3 gene mutation is present, the targeted therapy drug midostaurin (Rydapt®) could be administered together with chemotherapy. This drug is administered twice a day in tablet form. New treatments, using the same target molecule, are also being developed as clinical trials pending approval, if appropriate, and are generally administered orally, and in conjunction with intravenous chemotherapy.
- For patients whose leukaemic cells express the CD33 protein, the targeted therapy drug gemtuzumab ozogamicin (Mylotarg®) could be added to chemotherapy in favourable- or intermediate-risk patients.
- It should be noted that in some specific young patient profiles, the treatment of choice may not be 7+3 but VYXEOS (an adaptation of 7+3 approved for secondary leukaemias in relation to previous chemotherapy or with a history of myelodysplasia treatment-related acute myeloid leukaemia (t-AML) or AML with myelodysplasia-related changes) or the combination of venetoclax with azacitidine.
In cases where the leukaemia has spread to the nervous system, chemotherapy can also be administered into the cerebrospinal fluid through lumbar punctures (intrathecal chemotherapy).

It should be noted that the first phase of treatment usually requires a prolonged hospital stay of approximately 4 to 5 weeks.
 2. Post-remission or consolidation treatment phase
2. Post-remission or consolidation treatment phase
This phase aims to maintain complete remission and further consolidate it by destroying residual leukaemic cells (minimal residual disease) that could at any time begin to reproduce and cause a relapse.
For patients with acute myeloid leukaemia, there are three post-remission treatment options:
- Consolidation chemotherapy
- Consolidation chemotherapy followed by autologous bone marrow transplantation (patient’s own – autologous haematopoietic progenitor transplant)
- Consolidation chemotherapy followed by allogeneic bone marrow transplantation (matched, family or anonymous donor – allogeneic haematopoietic progenitor transplantation)
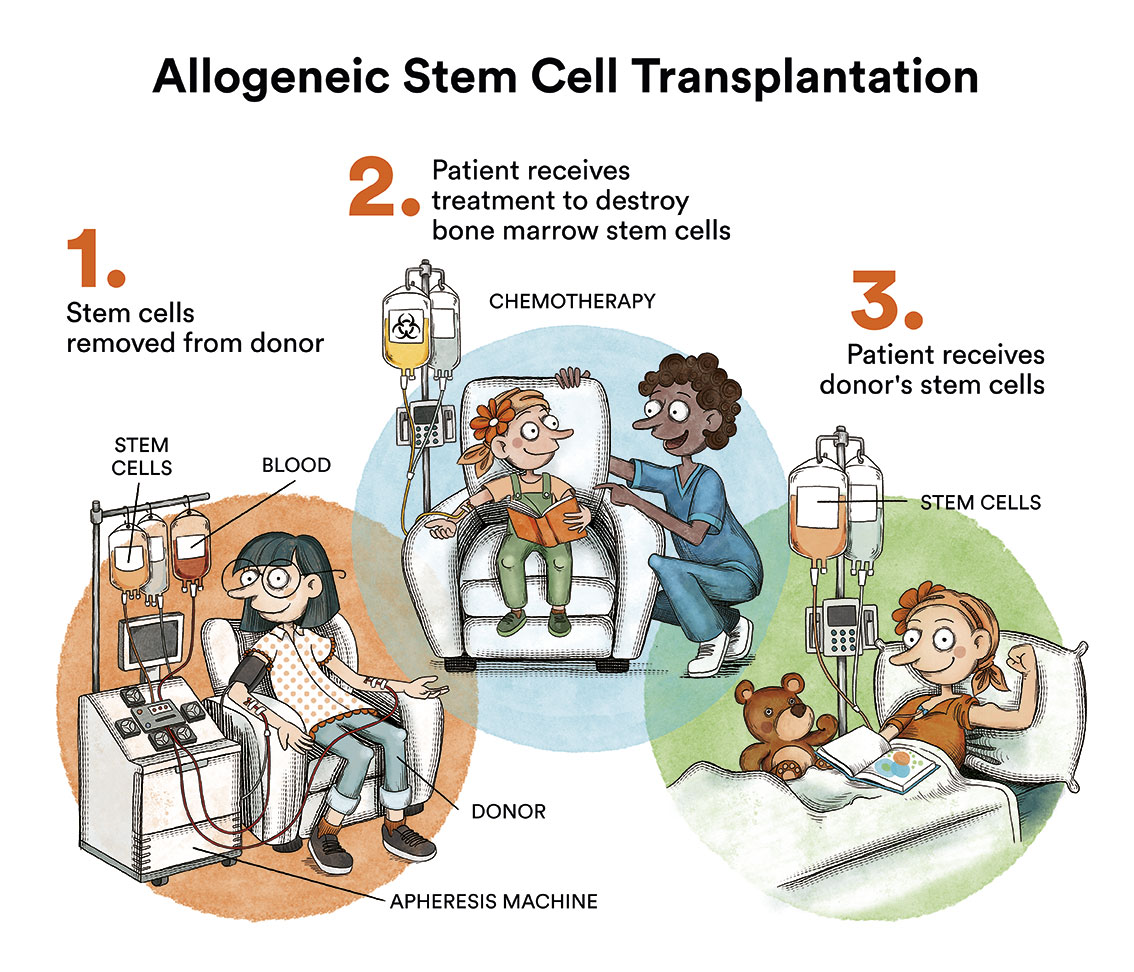
The first option (consolidation chemotherapy) is the one of choice in patients with a favourable prognosis (low risk of recurrence after intensive chemotherapy treatment) and no detectable minimal residual disease, which is studied through complementary laboratory tests (cytometry and molecular studies). There is insufficient evidence to recommend chemotherapy alone or chemotherapy followed by autologous bone marrow transplantation, so this will be decided on an individual patient basis.
Similarly, the optimal regimen to be used for consolidation and the number of cycles of chemotherapy to be administered are also not fully established. However, when a transplant is to be performed, 1 or 2 cycles of consolidation are usually administered, whereas when a transplant is not performed, a minimum of two cycles tends to be administered.
In some patients with subtypes of acute myeloid leukaemia that are considered to be intermediate or high risk (high risk of recurrence of the disease despite administering chemotherapy or after a first relapse), with ages below 70 years and without any additional significant medical issues, the indication is to perform a haematopoietic stem cell transplantation (bone marrow, peripheral blood or umbilical cord blood) from a compatible donor (allogeneic transplantation), ideally a histocompatible sibling or, failing that, a globally located unrelated volunteer donor or umbilical cord blood unit. There is also the option of transplantation with 50% compatible relatives (haploidentical) with very similar results to transplants with a higher compatibility.
In patients over 70 years of age, the decision whether or not to perform an allogeneic transplant is made on a case-by-case basis. In these cases, it is not the age itself that is important, but the patient’s general condition, tolerance to previous treatments, and the availability of a good donor. It applies equally to patients under 70 years of age, but with relevant comorbidities that make them ineligible for transplantation.
In elderly patients with previous poor functional status, given the expected poor tolerance to intensive chemotherapy, different therapeutic strategies are adopted. The most commonly used are low-dose chemotherapies or hypomethylating agents, such as Decitabine or Azacitidine, which aim to delay disease progression as much as possible while causing the least toxicity and prioritising quality of life.
The treatment of treatment-related acute myeloid leukaemia or leukaemia secondary to other haematological diseases does not differ much from other acute myeloid leukaemias, although the likelihood of achieving a sustained complete remission is lower due to greater resistance to chemotherapy. In these cases, if feasible, an allogeneic haematopoietic stem cell transplant is usually performed as the therapeutic approach with the best chance of cure.

Laura
Acute Myeloid Leukaemia
“When my little son was only 6 months old, I was diagnosed with acute myeloid leukemia. It was a very difficult time, but thanks to the professionals at the hospital, my children, and my brother’s bone marrow donation, I am alive today. I live better and happier.”
Specific subtypes: acute promyelocytic leukaemia and patients with Down Syndrome
Acute Promyelocytic Leukaemia
One of the leukaemias that has benefited most from an individualised therapeutic strategy is acute promyelocytic leukaemia (APL). In recent decades, thanks to scientific research, a substantial improvement in treatment has been achieved, from being a subtype of AML with a very poor prognosis to a disease that in most cases responds very well to treatment. This type of leukaemia is characterised by a translocation between chromosomes 15 and 17 [t(15:17)], which affects the retinoic acid receptor alpha (RARα or RARA) and confers a high sensitivity to treatment with holotransretinoic acid (ATRA). This is why this type of leukaemia is treated differently from other types of AML based on the use of ATRA.
Acute promyelocytic leukaemia (APL) accounts for 10-15% of all AML. The median age of these patients is 40 years.
The interval between the onset of the first symptoms and diagnosis of acute promyelocytic leukaemia is usually less than two months due to the acute nature of the disease. In addition to symptoms attributable to the anaemia caused by the red blood cell deficiency (feeling tired, weakness, dizziness, paleness), this leukaemia is characterised by significantly impaired coagulation. Bleeding is therefore usually present in up to 75% of patients in the form of bruising of the skin and mucous membranes; or bleeding in any other region, the most dangerous being in the central nervous system. In fact, bleeding accounts for 60% of deaths in this early stage of the disease, making early diagnosis of the disease with immediate initiation of ATRA and coagulation-enhancing factors very important. As with other leukaemias, a significant number of patients may experience fever due to intercurrent infections secondary to the lack of granulocytes. Occasionally, patients may also experience an enlargement of the lymph nodes, liver or spleen.
In addition to the basic blood and bone marrow studies (morphology, blood count and immunophenotype) to be performed in all leukaemia diagnoses, in acute promyelocytic leukaemia the rapid availability of cytogenetic and molecular biology results that will confirm clinical suspicion are of particular importance. Eighty percent of patients have the t(15;17) translocation and 99% have the PML-RARα gene, which allows for a safe diagnosis with important therapeutic implications.
In this type of acute leukaemia, initial treatment will also affect the risk of relapse. To calculate it, the most recent classifications use the number of leukocytes and platelets at the time of diagnosis.
In patients with favourable risk (leukocytes < 10,000/mm3 and platelets > 40,000/mm3), induction therapy uses ATRA, a member of the vitamin A family, in combination with arsenic trioxide (ATO). ATRA is also known as tretinoin (Vesanoid®), and is characterised by eliminating the PML/RARα abnormality and thus inducing promyelocytes to develop into mature cells (neutrophils).
In patients at adverse risk (leukocytes > 10,000/mm3), a chemotherapy agent, usually of the anthracycline class (daunorubicin or idarubicin), is also associated with the aim of reducing the number of disease cells in the blood more rapidly.
The side effects of this treatment, apart from those previously mentioned in relation to leukaemia (infections, haemorrhages), should be borne in mind, these include the possible appearance of a condition known as differentiation syndrome (precisely because of the immediate effect of ATRA on the differentiation of blasts), characterised by an increase in the number of leukocytes and fluid retention (manifested by the appearance of pulmonary infiltrates, effusions), as well as fever and hypotension, and whose treatment is based on corticosteroids and the temporary interruption of ATRA. On the other hand, the administration of ATO can affect the electrolytes in the blood and should therefore be associated with control analyses to correct them and avoid alterations in the heart rate.
Overall, this treatment achieves remission in approximately 80-90% of patients with acute promyelocytic leukaemia. After remission, consolidation cycles with ATRA and ATO are continued in varying numbers, followed by a prolonged phase of maintenance treatment in which the drugs used are usually unchanged.
In the case of a recurrence, treatment will be individualised for each patient, depending on their age, general condition and the drugs previously used. It is in this situation that a consolidation strategy involving allogeneic haematopoietic stem cell transplantation, usually autologous, can be considered. Allogeneic transplantation in APL is a very rare indication, to be considered in case of failure of autologous transplantation, which is exceptional.
With the new treatment strategies, survival is very high and relapses are very rare.
Patients with Down syndrome
People with Down syndrome have a 15 times higher risk of developing acute leukaemia. In acute myeloid leukaemia, the age of presentation is usually in children or young adults.
This group of patients has a high sensitivity to chemotherapy treatments and this has led to high cure rates. One of the main difficulties in achieving a cure is due to the high toxicity of some chemotherapy drugs and the high risk of infection. This is why different groups have been able to increase survival with adapted treatment protocols.
What are the chances of adult acute myeloid leukaemia patients being cured?
The prognosis of patients with acute myeloid leukaemia varies substantially depending on several factors including: age, history of previous chemotherapy or development of AML in individuals with previous haematological disorders such as myelodysplasia or myeloproliferative syndrome, the degree of initial leukocytosis, the presence of certain cytogenetic/molecular abnormalities, as well as the slowness in obtaining complete remission.
Thus, young patients with low-risk leukaemias (favourable group) have a cure probability of more than 75%, while intermediate-risk patients who receive an allogeneic transplant from an optimal donor have a cure probability of up to 65-70%. However, a patient with very high-risk leukaemias, especially if complete remission is not achieved with chemotherapy treatment, has very low chance of cure.
The specific case of acute promyelocytic leukaemia has an estimated 90% survival rate.
It is very important to note that survival rate statistics for people with acute myeloid leukaemia are an estimate and will be assessed on a case-by-case basis by the medical team.

Sara, 29 years old.
Acute Myeloid Leukaemia.
“At 17 years old I was diagnosed with acute myeloid leukaemia. It was a huge setback. My mother had just been diagnosed with breast cancer. In the summer of 2014, I underwent my bone marrow transplant from an unrelated donor located by the REDMO program of the Josep Carreras Foundation, and my mother also had surgery. After 9 years, although with quite a few limitations, I am studying to be a nurse.”
Monitoring
After completing treatment, it is very important to continue regular check-ups with the medical team, as well as with other specialists if necessary. Monitoring is carried out to confirm and aid physical recovery after treatment and to follow up and ensure early identification of possible long-term complications. Similarly, especially in the first months/years after the end of treatment, blood tests and bone marrow studies will be carried out to confirm the absence of recurrence of the disease. These controls are progressively spaced out. Long-term follow-up is recommended at least annually in order to early detection and treatment of any sequelae arising from treatment or leukaemia, should they appear.
Links of interest concerning medical issues relating to acute myeloid leukaemia in adults
- Acute lymphoblastic leukaemia in adults. American Cancer Society.
- Adult Acute Lymphoblastic Leukemia Treatment. National Cancer Institute
- Acute Lymphoblastic Leukemia. Leukemia & Lymphoma Society
- Acute lymphoblastic leukaemia. Bloodwise UK
Links of interest on other topics related to acute myeloid leukaemia in adults
TESTIMONIAL MATERIALS
You can order the booklets in paper format for free delivery in Spain by e-mail: imparables@fcarreras.es
BONE MARROW TRANSPLANT
- Bone Marrow Transplant Guide. Josep Carreras Foundation (content in Spanish)
- What is HLA and how does it work? Josep Carreras Foundation (content in Spanish)
- Graft-versus-Host Disease. Josep Carreras Foundation (content in Spanish)
- History of Bone Marrow Transplantation. Josep Carreras Foundation (content in Spanish)
- How is the search for an anonymous donor conducted? Josep Carreras Foundation (content in Spanish)
FOOD
- How to maintain a healthy diet during treatment? Josep Carreras Foundation (content in Spanish)
- Nutrition guide. Leukemia & Lymphoma Society
OTHER
- Ideas on what to take with me to the isolation chamber. Josep Carreras Leukaemia Foundation (content in Spanish)
- Travel tips for people with cancer. Josep Carreras Leukaemia Foundation (content in Spanish)
- Physiotherapy manual for haematological and transplant patients. Josep Carreras Leukaemia Foundation (content in Spanish)
- Prevention and treatment of oral mucositis. Josep Carreras Leukaemia Foundation (content in Spanish)
- Oral hygiene in oncohaematological patients. Josep Carreras Leukaemia Foundation (content in Spanish)
- Fertility manual: Suffering from blood cancer and becoming a parent. Josep Carreras Leukaemia Foundation (content in Spanish)
- Skin care in the oncohaematological patient. Josep Carreras Leukaemia Foundation (content in Spanish)
- Aesthetic Oncology Manual. Josep Carreras Leukaemia Foundation (content in Spanish)
- Leukaemia and sexuality. Josep Carreras Leukaemia Foundation (content in Spanish)
- 7 ways to wear a scarf. Josep Carreras Leukaemia Foundation (content in Spanish)
Links of interest: local/provincial or state entities that can provide you with resources and services specialised in leukaemia or cancer patients:
In Spain there is a large network of associations for haematological cancer patients that, in many cases, can inform you, advise you and even carry out certain procedures. These are the contacts of some of them by Autonomous Communities:
All these organisations are external to the Josep Carreras Foundation.
STATE
- CEMMP (Comunidad Española de Pacientes de Mieloma Múltiple)
- AEAL (ASOCIACIÓN ESPAÑOLA DE AFECTADOS POR LINFOMA, MIELOMA y LEUCEMIA)
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact with the nearest branch or call 900 100 036 (24h).
- AELCLES (Agrupación Española contra la Leucemia y Enfermedades de la Sangre)
- Josep Carreras Leukaemia Foundation
- FUNDACIÓN SANDRA IBARRA
- GEPAC (GRUPO ESPAÑOL DE PACIENTES CON CÁNCER)
- MPN España (Asociación de Afectados Por Neoplasias Mieloproliferativas Crónicas)
ANDALUCÍA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- ALUSVI (ASOCIACIÓN LUCHA Y SONRÍE POR LA VIDA). Sevilla
- APOLEU (ASOCIACIÓN DE APOYO A PACIENTES Y FAMILIARES DE LEUCEMIA). Cádiz
ARAGÓN
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- ASPHER (ASOCIACIÓN DE PACIENTES DE ENFERMEDADES HEMATOLÓGICAS RARAS DE ARAGÓN)
- DONA MÉDULA ARAGÓN
ASTURIAS
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- ASTHEHA (ASOCIACIÓN DE TRASPLANTADOS HEMATOPOYÉTICOS Y ENFERMOS HEMATOLÓGICOS DE ASTURIAS)
CANTABRIA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
CASTILLA LA MANCHA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
CASTILLA LEÓN
- ABACES (ASOCIACIÓN BERCIANA DE AYUDA CONTRA LAS ENFERMEDADES DE LA SANGRE)
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- ALCLES (ASOCIACIÓN LEONESA CON LAS ENFERMEDADES DE LA SANGRE). León.
- ASCOL (ASOCIACIÓN CONTRA LA LEUCEMIA Y ENFERMEDADES DE LA SANGRE). Salamanca.
CATALUÑA
- ASSOCIACIÓ FÈNIX. Solsona
- FECEC (FEDERACIÓ CATALANA D’ENTITATS CONTRA EL CÁNCER
- FUNDACIÓ KÁLIDA. Barcelona
- FUNDACIÓ ROSES CONTRA EL CÀNCER. Roses
- LLIGA CONTRA EL CÀNCER COMARQUES DE TARRAGONA I TERRES DE L’EBRE. Tarragona
- MielomaCAT
- ONCOLLIGA BARCELONA. Barcelona
- ONCOLLIGA GIRONA. Girona
- ONCOLLIGA COMARQUES DE LLEIDA. Lleida
- ONCOVALLÈS. Vallès Oriental
- OSONA CONTRA EL CÀNCER. Osona
- SUPORT I COMPANYIA. Barcelona
- VILASSAR DE DALT CONTRA EL CÀNCER. Vilassar de Dalt
VALENCIAN COMMUNITY
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- ASLEUVAL (ASOCIACIÓN DE PACIENTES DE LEUCEMIA, LINFOMA, MIELOMA Y OTRAS ENFERMEDADES DE LA SANGRE DE VALENCIA)
EXTREMADURA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- AFAL (AYUDA A FAMILIAS AFECTADAS DE LEUCEMIAS, LINFOMAS; MIELOMAS Y APLASIAS)
- AOEX (ASOCIACIÓN ONCOLÓGICA EXTREMEÑA)
GALICIA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- ASOTRAME (ASOCIACIÓN GALLEGA DE AFECTADOS POR TRASPLANTES MEDULARES)
BALEARIC ISLANDS
- ADAA (ASSOCIACIÓ D’AJUDA A L’ACOMPANYAMENT DEL MALALT DE LES ILLES BALEARS)
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
CANARY ISLANDS
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- AFOL (ASOCIACIÓN DE FAMILIAS ONCOHEMATOLÓGICAS DE LANZAROTE)
- FUNDACIÓN ALEJANDRO DA SILVA
LA RIOJA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
MADRID
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- AEAL (ASOCIACIÓN ESPAÑOLA DE LEUCEMIA Y LINFOMA)
- CRIS CONTRA EL CÁNCER
- FUNDACIÓN LEUCEMIA Y LINFOMA
MURCIA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
NAVARRA
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
BASQUE COUNTRY
- AECC (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER). Present is the different provinces and in many municipalities. Contact the nearest branch.
- PAUSOZ-PAUSO. Bilbao
AUTONOMOUS CITIES OF CEUTA AND MELILLA
- AECC CEUTA (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER)
- AECC MELILLA (ASOCIACIÓN ESPAÑOLA CONTRA EL CÁNCER)
Support and assistance
We also invite you to follow us through our main social media (Facebook, Twitter and Instagram) where we often share testimonies of overcoming this disease.
If you live in Spain, you can also contact us by sending an e-mail to imparables@fcarreras.es so that we can help you get in touch with other people who have overcome this disease.
* In accordance with Law 34/2002 on Information Society Services and Electronic Commerce (LSSICE), the Josep Carreras Leukemia Foundation informs that all medical information available on www.fcarreras.org has been reviewed and accredited by Dr. Enric Carreras Pons, Member No. 9438, Barcelona, Doctor in Medicine and Surgery, Specialist in Internal Medicine, Specialist in Hematology and Hemotherapy and Senior Consultant of the Foundation; and by Dr. Rocío Parody Porras, Member No. 35205, Barcelona, Doctor in Medicine and Surgery, Specialist in Hematology and Hemotherapy and attached to the Medical Directorate of the Registry of Bone Marrow Donors (REDMO) of the Foundation).
Become a member of the cure for leukaemia!


