Childhood acute lymphoblastic leukaemia, type T
Leukaemia is the most common cancer among children. It affects more than 350 children a year in our country, a third of which are under 4 years of age. Leukaemia accounts for 30% of all paediatric cancers, with B-acute lymphoblastic leukaemia being the most common form among children (80% of cases)
The information provided on www.fcarreras.org is intended to support, not replace, the relationship that exists between patients/visitors to this website and their physician.

Information endorsed by… ![]()
What is leukaemia, bone marrow and what are the types of blood cells?
Leukaemia is a type of blood cell and bone marrow cancer. See section Leukaemia, bone marrow and blood cells.
What is T-acute lymphoblastic leukaemia?
 Acute lymphoblastic leukaemia (ALL) is a type of cancer in which the bone marrow produces too many immature lymphocytes (a type of white blood cell), known as lymphoblasts. These cells prevent the growth of other blood cells. ALL is the most common cancer in children, and most diagnoses are B-acute lymphoblastic leukaemia (80% of leukaemia cases in children). T-lymphoblastic leukaemia is a rare, heterogeneous subtype of the disease that affects only a few children per year in Spain. It accounts for 10-15% of ALL cases in children.
Acute lymphoblastic leukaemia (ALL) is a type of cancer in which the bone marrow produces too many immature lymphocytes (a type of white blood cell), known as lymphoblasts. These cells prevent the growth of other blood cells. ALL is the most common cancer in children, and most diagnoses are B-acute lymphoblastic leukaemia (80% of leukaemia cases in children). T-lymphoblastic leukaemia is a rare, heterogeneous subtype of the disease that affects only a few children per year in Spain. It accounts for 10-15% of ALL cases in children.
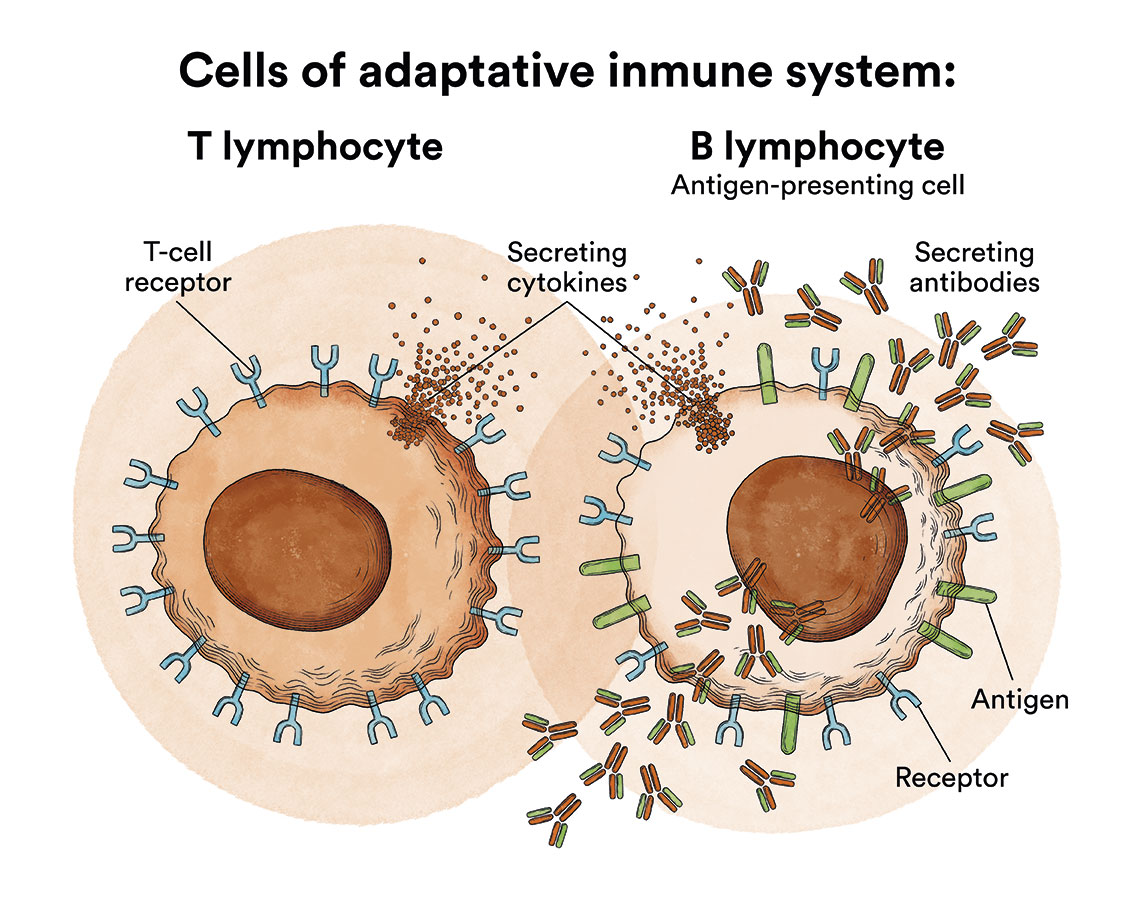
Lymphocytes are a type of leukocyte or white blood cell and can be of two types, B and T. Depending on whether the lymphoblasts originate from B or T cells, ALL will be referred to as B-ALL or T-ALL.
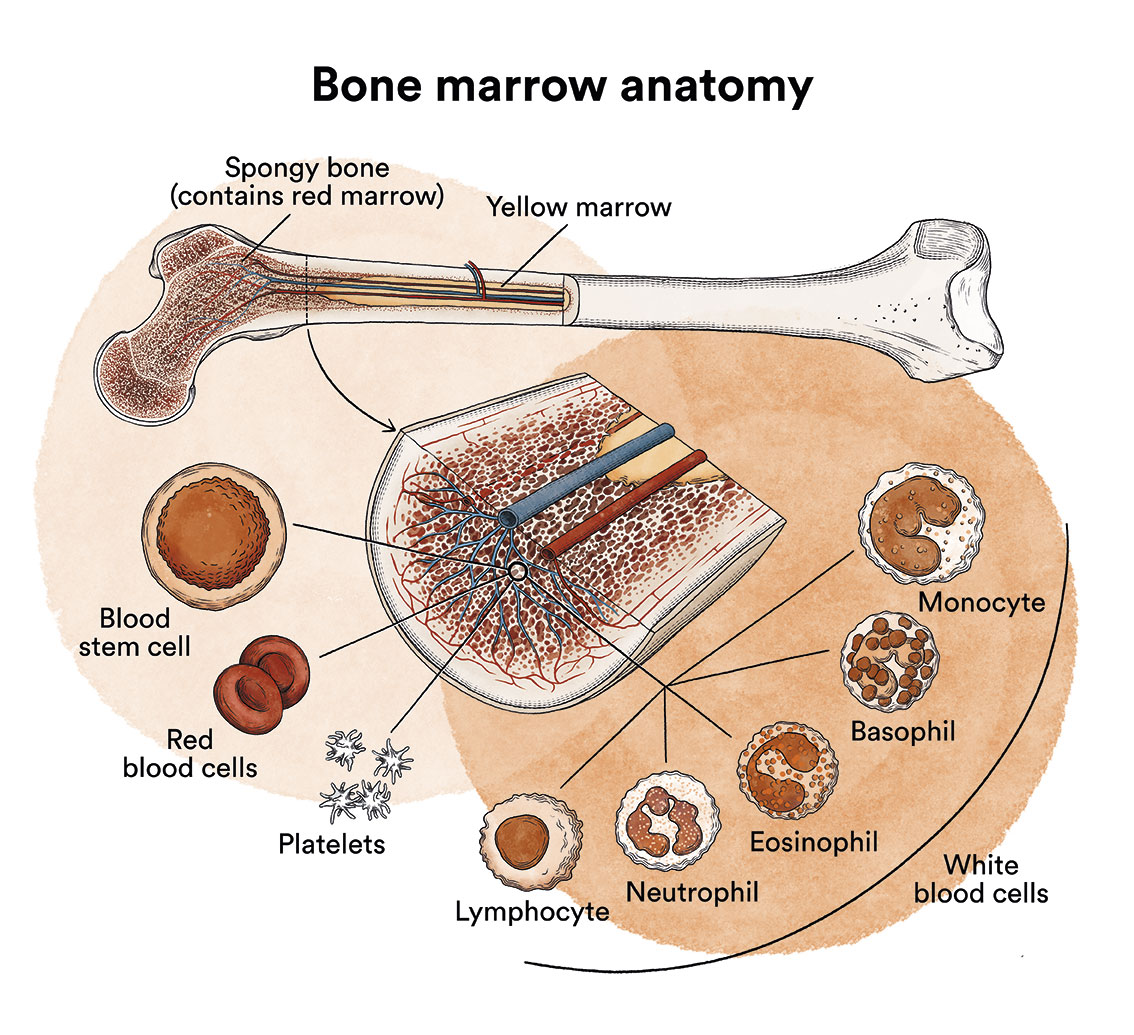
As we have seen in ‘Leukaemia, bone marrow and blood cells’, the bone marrow produces blood stem cells (immature cells) that will eventually develop into mature blood cells. A blood stem cell becomes a myeloid stem cell or a lymphoid stem cell.
A myeloid stem cell develops into one of the three types of mature blood cells:
- Red blood cells, which carry oxygen to other tissues and
organs in the body. - Granulocytes, white blood cells that help fight infection
and other diseases. - Platelets, which help blood to clot when
a blood vessel ruptures.
- Red blood cells, which carry oxygen to other tissues and
A lymphoid stem cell develops into a lymphoblast and later into one of the three types of lymphocytes (white blood cells):
- B lymphocytes, which produce antibodies to help fight infections in the body.
- T lymphocytes, which help B lymphocytes produce antibodies to fight infection.
- Natural cytolytic lymphocytes or NK lymphocytes (natural killer), a type of immune cell that contains enzymes which can destroy tumour cells or virus-infected cells.
In children affected by T-acute lymphoblastic leukaemia, there are too many stem cells that transform into T lymphoblasts.
What are the causes of T-acute lymphoblastic leukaemia?
The exact cause of T-ALL is unknown. Research in this field is important and the genetic changes that occur in leukaemic cells are becoming known.
However, in most children with T-ALL, the triggers or factors that led to the leukaemia are unknown.
In a minority of children there are some genetic diseases that predispose them to the development of acute lymphoblastic leukaemia. This is the case for children with Down syndrome or other syndromes such as Li-Fraumeni syndrome, ataxia-telangiectasia, Fanconi anaemia and others. Other factors that increase the likelihood of developing leukaemia may include exposure to radiation prior to birth (X-rays) or having received chemotherapy for another cancer.
Siblings of a child with leukaemia, compared to other children, have a small increased risk of developing leukaemia. However, this risk is very low, less than 1 per thousand.
Leukaemia, like other cancers, is not contagious. See section Leukaemia, bone marrow and blood cells.
What are the symptoms of T-acute lymphoblastic leukaemia?
In T-acute lymphoblastic leukaemia, the production of normal blood cells is disrupted by the growth of leukaemic cells in the bone marrow.
This can lead to:
- Tiredness and paleness (due to anaemia).
- Appearance of purple and small pink patches on the skin (petechiae) or other bleeding due to low platelet count.
- Fever and infections that do not progress well (due to leukocyte malfunction).
- Joint and bone pain or lameness (due to leukaemia cells having invaded the bone marrow).
- Adenopathies or enlarged lymph nodes (due to the occupancy of leukaemic cells in the lymphatic system).
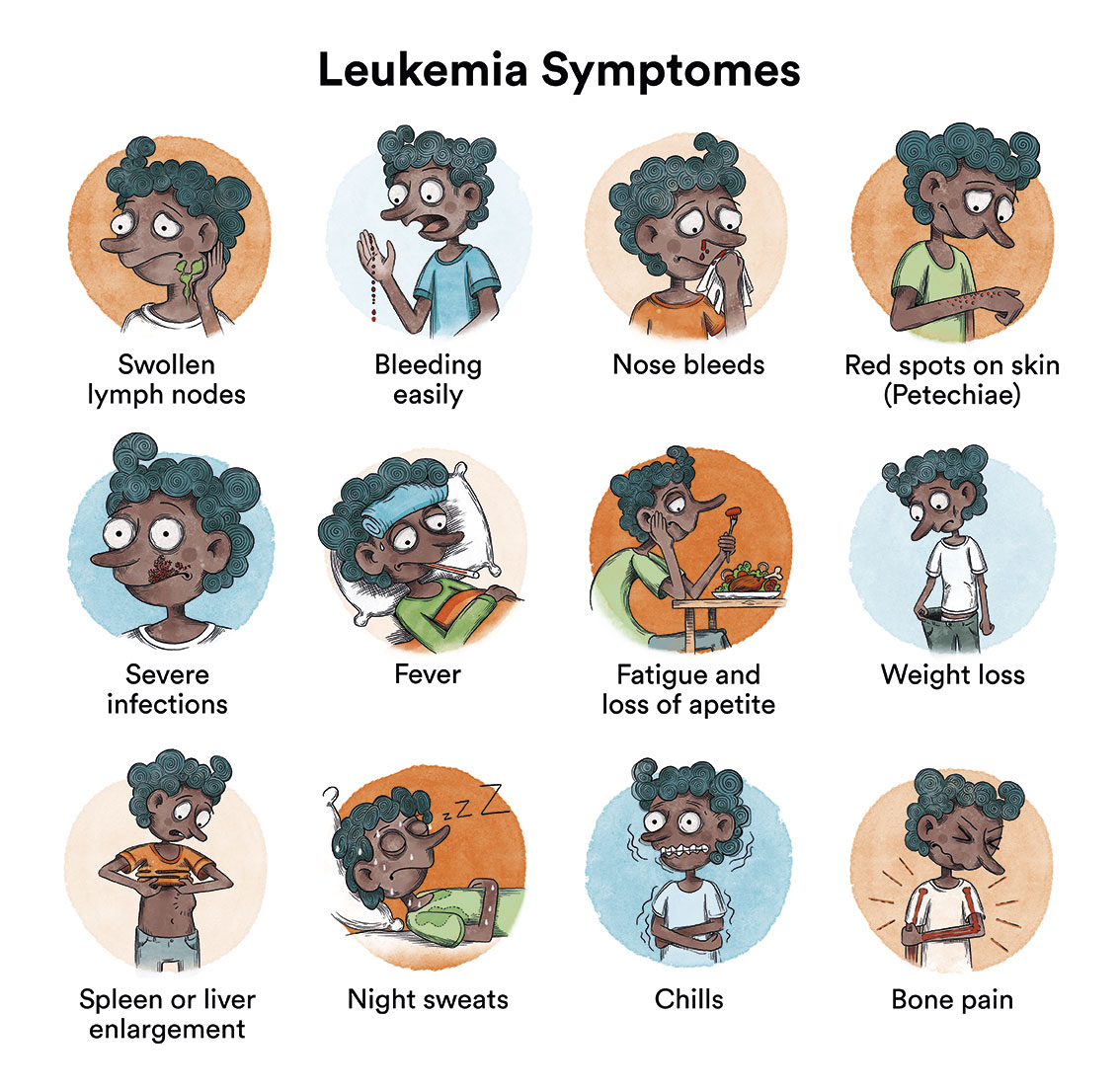
At the onset of the disease, all these symptoms can be very similar to those of a virus infection. When symptoms continue for more than 2-4 weeks, a diagnosis can be obtained in most cases. As these symptoms are not specific or exclusive to leukaemia, it is very common for the patient to have visited a doctor several times before being diagnosed. Generally, this does not influence the child’s curative options.
How is T-acute lymphoblastic leukaemia diagnosed?
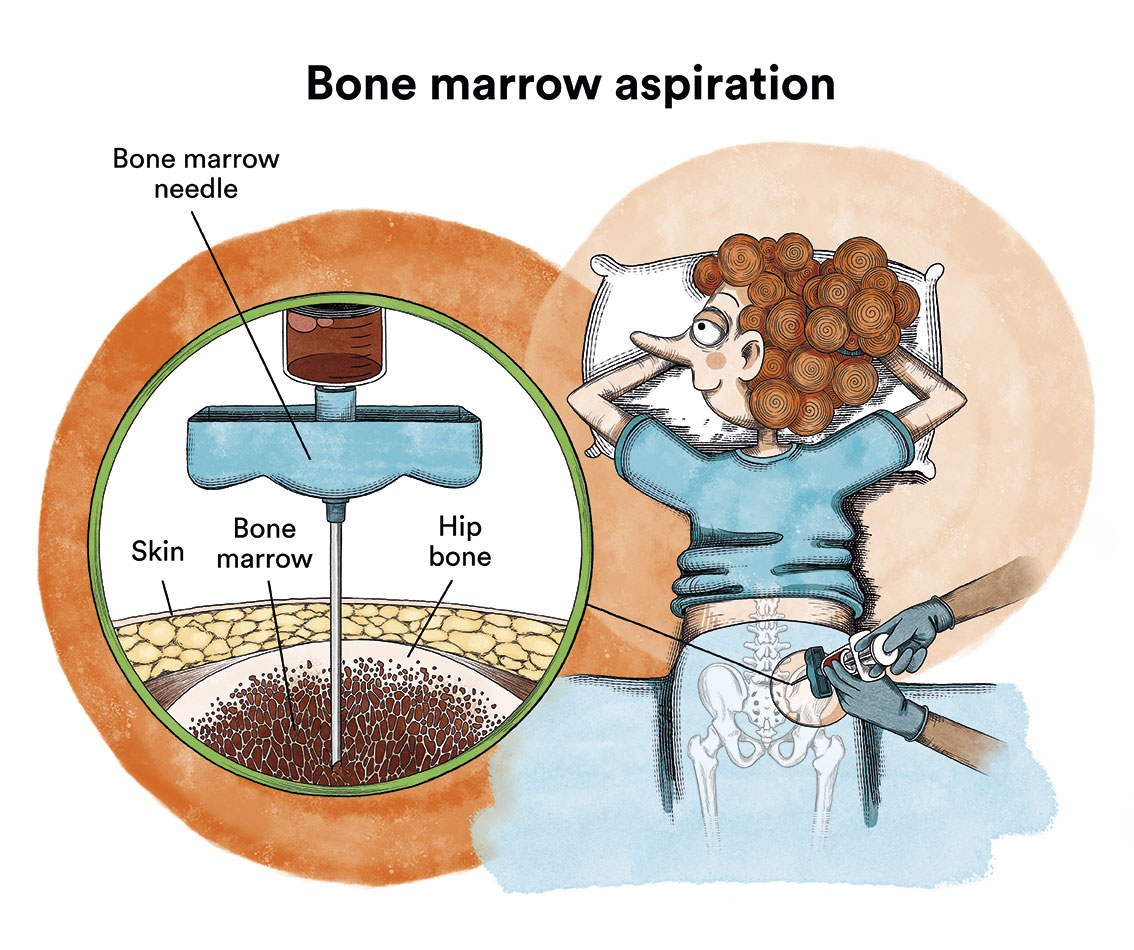
Generally, if a child presents the characteristic symptoms, leukaemia can be diagnosed with a blood test. When the blood is examined under a microscope, leukaemic cells will be visible. Sometimes, at the beginning of the disease, these cells are not seen in the blood and leukaemia is suspected due to the symptoms and some alterations in the blood tests. The diagnosis is confirmed by a bone marrow puncture (iliac crests) and analysis of the cells obtained.
What is the treatment for T-acute lymphoblastic leukaemia in children?
Treatment of newly diagnosed childhood T-cell acute lymphoblastic leukaemia (T-ALL) in the remission induction, consolidation and intensification and maintenance phases always includes chemotherapy combined with other drugs. Children with T-ALL are given more anticancer drugs and at higher doses than children in the standard risk group who are newly diagnosed, although the same drugs and chemotherapy schedules are currently used as for B-ALL.

They are administered intrathecal and systemic chemotherapy to prevent or treat the spread of leukaemic cells to the brain and spinal cord. Sometimes, radiotherapy to the brain is also sometimes administered.
The goal of treatment for acute lymphoblastic leukaemia is to eliminate the leukaemic cells to allow the bone marrow to return to its normal functioning.
The child’s team of haematology doctors will decide on the best treatment for them, taking into consideration many factors, including:
- The type of ALL the child has (B or T)
- The child’s age and the number of leukocytes at diagnosis
- The genetic characteristics of the leukaemic cells
- The child’s response to treatment
The treatment of ALL follows treatment protocols developed by paediatric haematology and oncology specialists and is based on the results of other protocols (from national and international groups).
Basically, chemotherapy is the main treatment the child will receive. It involves the use of drugs that kill cancer cells by preventing them from reproducing.
When chemotherapy is administered intravenously, to avoid repeatedly puncturing a vein, specialists implant a special device called a catheter (content in spanish). The catheter is inserted into a large vein, which allows for the administration of all types of medication, as well as for drawing blood for blood tests, thus avoiding the child from enduring repeated needle punctures.
There is a type of catheter, called a port-a-cath (content in spanish), which is attached to a round plastic or metal reservoir under the skin of the chest. The port-a-cath is very practical for children because it is under the skin and does not allow the child to pull it out, it is less likely to become infected than other types of catheters and it allows the child to bathe.
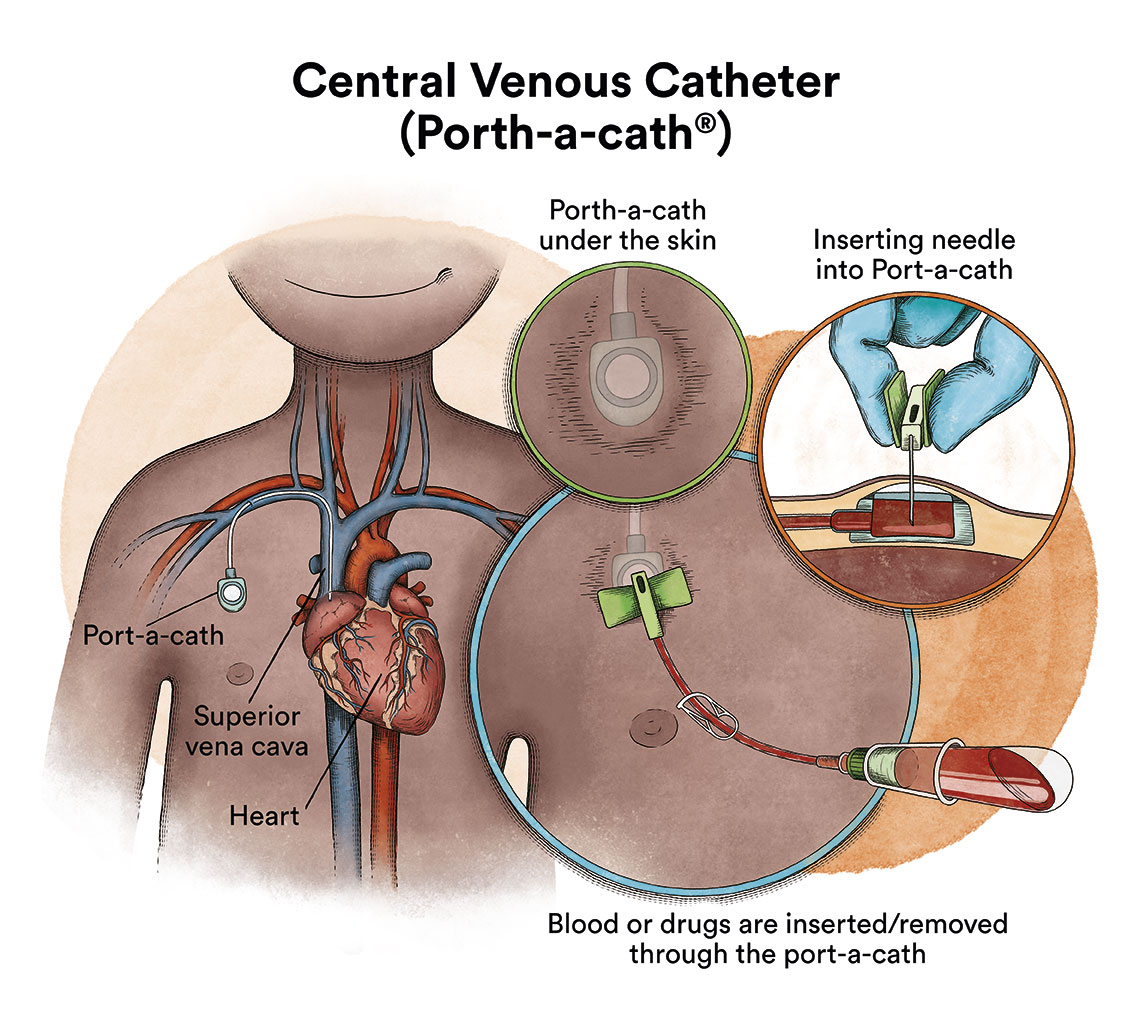
If the chemotherapy is administered through a venous catheter, it reaches almost all the cells of the body via the blood. However, most chemotherapy drugs do not fully reach the cerebrospinal fluid that bathes the brain and spinal cord. This means that there are leukaemic cells that can survive in this fluid. In order to prevent leukaemic cells that reach the cerebrospinal fluid from surviving and causing a future relapse that will affect the nervous system, chemotherapy must be administered directly into the cerebrospinal fluid, via lumbar punctures (intrathecal chemotherapy). In a minority of children who, due to the characteristics of their ALL, are known to have a higher risk of relapse in the nervous system, this treatment can be complemented with cranial radiotherapy.
Chemotherapy treatment can cure up to 85-90% of children with T-ALL. When no leukaemia cells are seen in the bone marrow or elsewhere, the patient is said to be in complete remission. The prognosis of T-cell ALL is not greatly affected by the age of the patient. There is a special subtype of the disease with a poor prognosis that is currently being researched. These are the Early pre-T ALL, which accounts for 12% of all T-ALL), for which chemotherapies with mixed activity (myeloid and lymphoid) as well as new drugs are being evaluated in clinical trials. In this case, a bone marrow transplant is recommended for all patients. Further studies are currently needed to establish therapeutic recommendations for these cases in childhood. In the current protocol for early pre-T (TOTAL XVII St Jude Children’s Research Hospital) treatment is intensified and the drug ruxolitinib is added.
In general, 15-20% of children can relapse from leukaemia. Most relapses occur in the first 5 years after diagnosis, especially in the first 2 years after the end of treatment. In cases of relapse, chemotherapy is re-administered and, depending on the case, a bone marrow transplant is performed (see ALL RELAPSE). There are also new treatment modalities for relapsing children (see NEW TREATMENTS)
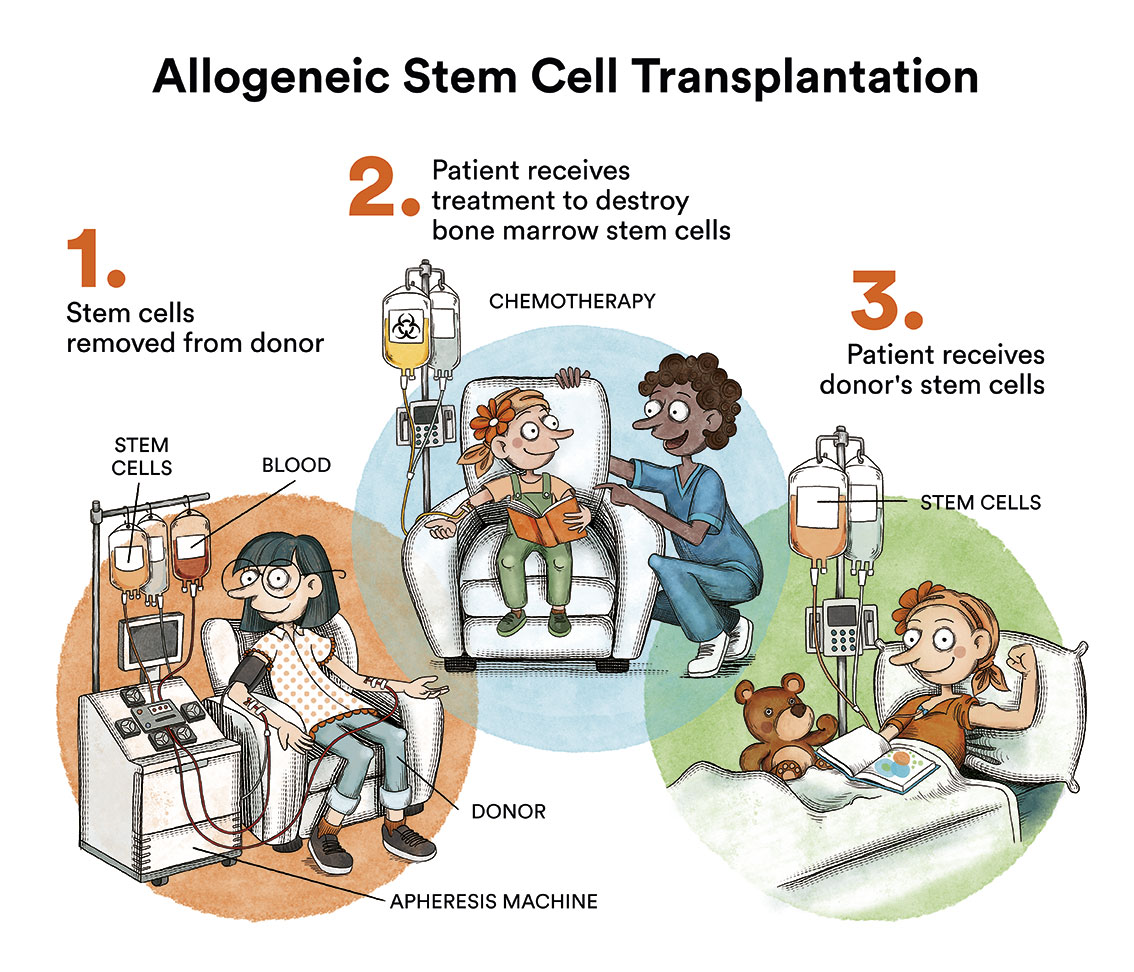
Transplantation is reserved for cases that are more difficult to cure with chemotherapy alone as it is a more intensive treatment with more risks and possible sequelae.
In children who have relapsed or in those who, due to the characteristics of their leukaemia, we know have a very high risk of relapse, a bone marrow transplant is indicated. This is performed once a new complete remission has been achieved with intensive chemotherapy or other treatment modalities. With transplantation, the chances of cure may increase.
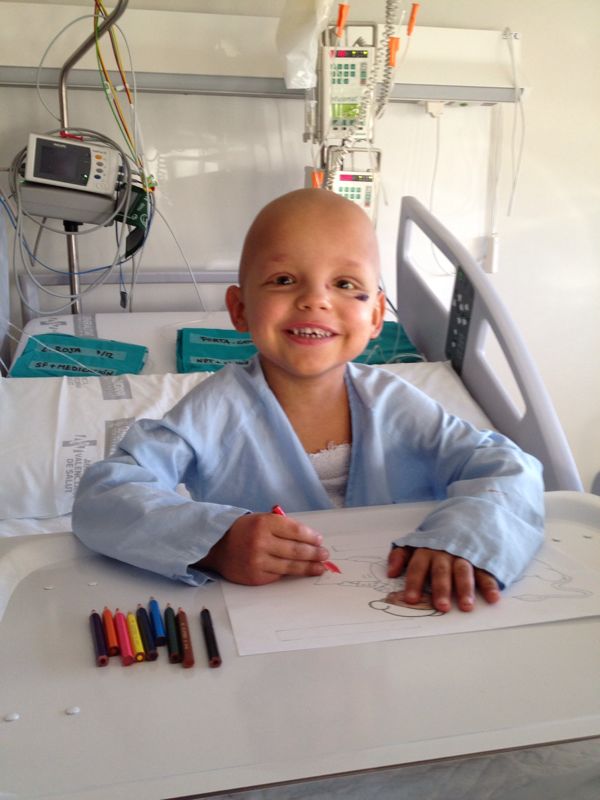
Jorge
Acute lymphoblastic leukaemia.
“Jorge was diagnosed with acute lymphoblastic leukemia when he was very little. In his house they always said that Jorge had arrived ‘as a gift’ when his parents wanted to have a little sister for his eldest son, Pablo. And so, the twins arrived: Lucía and Jorge. What they discovered over time is that the real gift had been Lucía, since she was 100% compatible with Jorge, who needed a bone marrow transplant to be cured. The transplant was not easy and, obviously, they were very scared. But as Fran, his mother, says, Jorge crossed the finish line. And years later he can tell that superheroes like him have already won her the game in life once. Now he must be and do what he sets out to do. It’s been 10 years since his transplant and Jorge is doing great.”
What are the chances of T-acute lymphoblastic leukaemia child patients being cured?
In T-phenotype ALL (T-ALL, 15% of cases) prognostic factors are not well defined. The 4- to 5-year disease-free survival rates of patients with T-ALL are on par with those of B-ALL (around 80%) but with more intensive treatment. The chances of cure may vary according to age (best prognosis between 1 and 9 years), the number of leukocytes at diagnosis (best if low) and the biological characteristics of the leukaemia (depending on the type of leukaemia and the genetic alterations in the cancer cells). The most important factor influencing prognosis is the treatment given to the child and the child’s response to this treatment. To assess this response, a series of bone marrow punctures will be performed throughout the course of treatment.

What happens if the child relapses?
If the child suffers a relapse of acute lymphoblastic leukaemia, the chances of cure and treatment will depend on several factors, mainly:
- of the time between diagnosis and relapse, the longer the better
- whether it is acute lymphoblastic leukaemia type B or type T
- the location of the relapse (bone marrow or other sites such as the nervous system, testicles or other)
- the child’s response to treatment of the relapse
Treatment will usually consist of more intensive chemotherapy than that initially received and often, bone marrow transplantation.
In recent years, in addition to chemotherapy, there are new treatments that can replace or complement chemotherapy, such as immunotherapy and treatments directed against a molecular target (see NEW TREATMENTS).
New treatments for T-acute lymphoblastic leukaemia
In recent years there has been a revolution in the treatment of cancer and specifically B-acute lymphoblastic leukaemia. Among the new treatments available to treat B-ALL are the treatments known as precision medicine and immunotherapy, especially CAR-T immunotherapy.
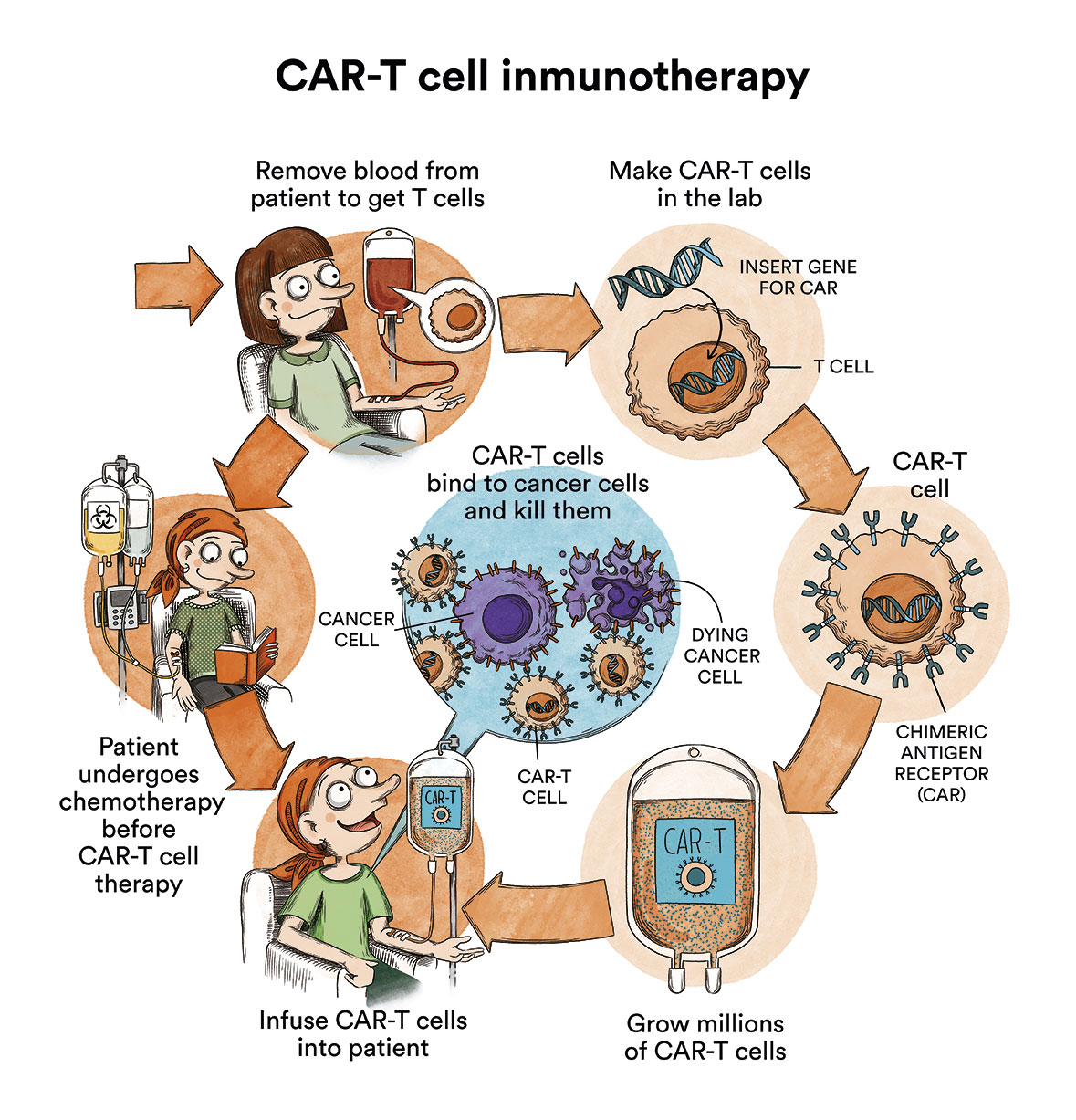
 Despite this, progress is also being made in the form of clinical trials for T-ALL.
Despite this, progress is also being made in the form of clinical trials for T-ALL.
In the case of relapse, nelarabine is currently the only approved drug, although its efficacy is limited and it can have serious side effects. Although clinical trials with target therapies (venetoclax, navitoclax, JAK inhibitors, among others), associated or not with chemotherapies, are currently being evaluated.
Immunotherapy treatments are cancer treatments that help the immune system fight cancer. The patient’s own immune system is boosted to help their body fight infections and other diseases. Immunotherapy can be used alone or in combination with chemotherapy or other cancer treatments.
Among other cell therapies, treatment with CAR-T cells has shown promising results in children with B-type ALL. CAR-T lymphocytes are lymphocytes that are extracted from the patient and genetically modified to recognise and destroy the leukaemic cell. See CAR-T imunotherapy or how to re-engineer your own cells to attack cancer In patients with T leukaemia, the immunotherapy strategy has the complexity and disadvantage that the T cells themselves (the ones that are modified to attack the cancer) are the ones that are sick. Nevertheless, CAR-T strategies are being developed, still in the form of clinical trials, against CD7, CD5 or CD1a antigens for cortical thymic T-ALL). These may show promising results over time.
Currently, in order to be able to perform CAR-T immunotherapies in patients with T-type lymphoblastic leukaemias, a therapy called STAb is being developed. This therapy is based on the secretion of a special type of antibody called bispecific that can recognise two targets, one on the tumour cell and one on the T-cell. In this way, the bispecific antibodies create a kind of artificial bridge that brings therapeutic T-cells into contact with tumour cells, facilitating the elimination of the latter and keeping healthy T-cells safe.
This distinction is essential in order to treat T-cell acute lymphoblastic leukaemia. In B-cell acute lymphoblastic leukaemia (B-ALL), CAR-T cells recognise a single target and destroy both diseased and healthy B-cells, although these patients can live a normal life thanks to the regular supply of immunoglobulins – antibodies – obtained from healthy donors.
In T-ALL, it is more complicated to administer CAR-T therapy because the cells used to fight the tumour – T-lymphocytes – are the same ones that are diseased and their use can lead to a state of immunodeficiency that is incompatible with life. In addition, there is no replacement therapy available as there is for B-cell leukaemias.
Monitoring
After completing the treatment, the child will have regular check-ups performed by their haematologist and by other specialists where necessary. Monitoring is carried out to assess possible relapse and to follow-up and treat possible long-term complications. These checks are progressively spaced out until they are carried out once a year. Long-term follow-up is recommended at least annually in order to early detection and treatment of any sequelae arising from treatment or leukaemia, should they appear.
Recommendations and other practicalities
Here are some general recommendations that answer some of the most frequently asked questions by parents of children with leukaemia:
- Will their hair fall out? When? Should we cut it?
With the chemotherapy they will receive to treat the leukaemia, their hair will fall out. This usually happens 2-3 weeks after the start of chemotherapy. If the child has long hair, it is more appropriate to cut it short before it starts to fall out. It is neither necessary, nor psychologically desirable, to cut it during the first days of admission. Nor does this fact need to be explained to the child straight away. However, it is important to address this issue with the child before their hair starts to fall out. Hair regrows 2-4 weeks after starting the maintenance phase of treatment, where chemotherapy is less intense.
- Hygiene
Since the child’s defences against infection are weakened (due to the illness itself and also due to the treatment administered), it is advisable to maintain adequate hygiene of the child’s body, the hospital room and the family home, including the child’s toys.
It is advisable to avoid dusty toys and cardboard boxes. Food should also not be stored outside the refrigerator. Plants are forbidden in the room, as there are fungal spores in the soil.
Tidiness facilitates cleaning by hospital cleaning staff.
- Visits
It is advisable to reduce the number of visitors in the child’s room, as they may carry infections. It is recommended that there are no more than 2 companions in the room and that they wash their hands before entering. If any of the visitors has an infectious process (cold, conjunctivitis…) it is preferable that they do not visit.
In the case of a parent or other caregiver whose attention cannot be dispensed with, they should wear a mask and wash their hands before coming into contact with the child.
- Food
A child receiving intensive chemotherapy treatment should be provided with a varied diet. When the white blood cell count is low, it is advisable to avoid raw foods that cannot be peeled (e.g. lettuce, strawberries, raw tomatoes).
Sometimes chemotherapy can reduce hunger, or even cause nausea. During the days of chemotherapy, it is not advisable to force your child to eat, as this can be counter-productive.
On the other hand, the corticosteroids (prednisone and dexamethasone) that the child will be administered during some phases of treatment can greatly increase their appetite, even causing anxiety. While they may be allowed to eat more than the meals served in the hospital, they should not be allowed to eat unlimited food, as they will often not tolerate it well and it can cause stomach pain.
Links of interest concerning medical issues relating to acute lymphoblastic leukaemia in children
Childhood Acute Lymphoblastic Leukemia Treatment. National Cancer Institute
Childhood acute lymphoblastic leukaemia (ALL). Cancer Research UK
Childhood acute lymphoblastic leukaemia (ALL). Blood Cancer UK
Acute Lymphoblastic Leukemia (ALL). St Jude Children’s Research Hospital
Links of interest on other topics related to acute lymphoblastic leukaemia in children
CHILDHOOD LEUKAEMIA MATERIAL
- Babies also have leukaemia. Josep Carreras Leukaemia Foundation. (content in Spanish)
- Childhood leukaemia. The little unstoppable ones. Josep Carreras Leukaemia Foundation. (content in Spanish)
- Medulin cut-out set. Josep Carreras Leukaemia Foundation. (content in Spanish)
The Josep Carreras Foundation has a story “The tough baby” aimed at children or siblings suffering from leukaemia. It is especially aimed at children up to the age of 6. If you want to order it, please send us an e-mail to imparables@fcarreras.es.
BONE MARROW TRANSPLANT
- Bone Marrow Transplant Guide. Josep Carreras Leukaemia Foundation.
- What is HLA and how does it work? Josep Carreras Leukaemia Foundation. (content in Spanish)
- Graft-versus-Host Disease Josep Carreras Leukaemia Foundation. (content in Spanish)
- History of Bone Marrow Transplantation. Josep Carreras Leukaemia Foundation. (content in Spanish)
- How is the search for an anonymous donor conducted? Josep Carreras Leukaemia Foundation. (content in Spanish)
- Care guide for transplanted children. TransplantCHild.
- Stem cell transplantation: a colouring book. Leukaemia and Lymphoma Society.
SUPPORT MANUALS
- How to deal with leukaemia and lymphoma in children? Leukemia & Lymphoma Society.
- LIVING BY LEARNING. Action protocol for students with cancer AFANION.
- Support guide for parents of children with cancer ASION.
- A guide for young people and adolescents with cancer ASION.
- Pupils with cancer. A guide for teachers ASION.
- The importance of parental behaviour when a child has cancer ASION.
- My child has cancer. What do I do? FARO.
FOOD
- How to maintain a healthy diet during treatment? Josep Carreras Leukaemia Foundation. (content in Spanish)
- “Bon appetit”. Dietary advice during treatment AFANION.
- ‘Jabel’s magic recipes’. Isabel Rojas Murcia, Carolina Mangas Gallardo.
OTHER
- Information on the long-term and late effects of treatment for leukaemia/lymphoma in children Leukemia & Lymphoma Society.
- My sibling has cancer Josep Carreras Leukaemia Foundation. (content in Spanish)
- School in a hospital Josep Carreras Leukaemia Foundation. (content in Spanish)
- Educating illusions. Guide for psycho-educational intervention in children and adolescents with cancer FARO.
- Cancer in adolescents Josep Carreras Leukaemia Foundation. (content in Spanish)
- ‘Leukaemia and adolescents’ documentary Josep Carreras Leukaemia Foundation.
- ‘Babies also have leukaemia’ documentary Josep Carreras Leukaemia Foundation.
- 7 ways to wear a scarf Josep Carreras Leukaemia Foundation. (content in Spanish)
- ‘Princess Luzie and the chemo knights’ story ASPANAFOA.
- ‘Let’s go to chemotherapy’ story.
- ‘Let’s go to radiotherapy’ story.
- ‘Gasparín Super Quimio’ story Spanish Federation of Parents of Children with Cancer.
- ‘Charlie Brown and leukaemia’ story.
- ‘Toby and the flying machine’ story.
- ‘The fairy of the stars’ story AECC.
- ‘Lina the little swallow’ story Osakidetza.
Useful links: local entities (resources and services)
ANDALUCÍA
ARAGÓN
ASTURIAS
CASTILLA LA MANCHA
CASTILLA LEÓN
CATALUÑA
VALENCIAN COMMUNITY
EXTREMADURA
GALICIA
BALEARIC ISLANDS
CANARY ISLANDS
LA RIOJA
MADRID
- AAA (asociación de adolescentes y Adultos Jóvenes con Cáncer)
- ASION
- FUNDACIÓN CAICO
- FUNDACIÓN ALADINA
- FUNDACIÓN UNOENTRECIENMIL
MURCIA
NAVARRA
BASQUE COUNTRY
Support and assistance
We also invite you to follow us through our main social media (Facebook, Twitter and Instagram) where we often share testimonies of overcoming this disease.
If you live in Spain, you can also contact us by sending an e-mail to imparables@fcarreras.es so that we can help you get in touch with other people who have overcome this disease.
* In accordance with Law 34/2002 on Information Society Services and Electronic Commerce (LSSICE), the Josep Carreras Leukemia Foundation informs that all medical information available on www.fcarreras.org has been reviewed and accredited by Dr. Enric Carreras Pons, Member No. 9438, Barcelona, Doctor in Medicine and Surgery, Specialist in Internal Medicine, Specialist in Hematology and Hemotherapy and Senior Consultant of the Foundation; and by Dr. Rocío Parody Porras, Member No. 35205, Barcelona, Doctor in Medicine and Surgery, Specialist in Hematology and Hemotherapy and attached to the Medical Directorate of the Registry of Bone Marrow Donors (REDMO) of the Foundation).
Become a member of the cure for leukaemia!


